Inflammatory Bowel Disease (IBD) Assays
If you are developing new IBD therapeutics, REPROCELL offers a selection of human fresh tissue and bioengineered tissue models to support your drug development pipeline. Whether you are looking to identify lead compounds or analyze clinical data, our scientists can help you at any stage of drug discovery.
At REPROCELL, we offer test systems in both human fresh intact IBD tissues donated by patients with UC or Crohn's disease, and leading-edge bioengineered tissue models of IBD based on our Alvetex® platform. Our experts can guide your selection of the appropriate models depending on your needs and have extensive experience using IBD fresh tissues and creating 3D tissues.
Explore our IBD assays
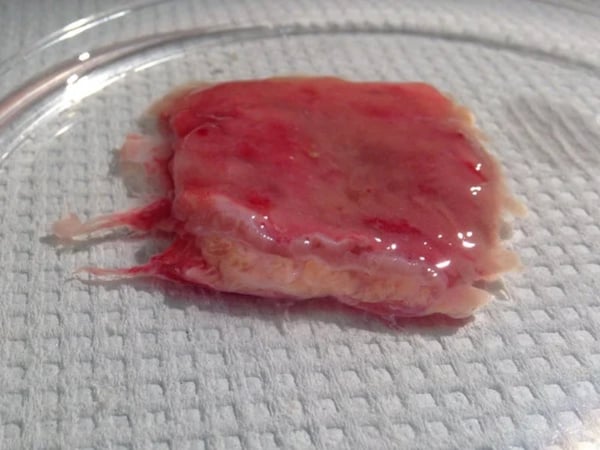
Anti-inflammatory Drug Responses in Human Fresh IBD Tissues
Our IBD culture model assesses the anti-inflammatory properties of your lead compounds in explants from patients with ulcerative colitis or Crohn's disease. More than 40 inflammatory mediators can be measured in this fresh tissue assay, which maintains the dynamic pharmacology and architecture of the intact intestine for up to 24 hours.

Pharmacology-AI: IBDiscovery
If you are looking to obtain actionable insights from your IBD patient data, Pharmacology-AI makes processing and analyzing large datasets easier. This machine learning (ML) powered platform identifies the key features driving biomarker levels, drug response, or clinical outcomes; streamlining your precision medicine strategies.
What IBD researchers say about working with us
“REPROCELL’s IBD culture studies have dynamically informed the potential of the HT-003 therapeutic platform, including focusing future development on lead candidate molecules with optimized target responses for ulcerative colitis and Crohn’s disease. The volume and quality of data we have been able to gather from these studies in a short amount of time is remarkable”
— Dr. Stefanie Johns, Chief Scientific Officer of Hoth Therapeutics.
Limitations of current IBD models in animals
Inflammatory bowel disease (IBD) is a chronic illness affecting the gastrointestinal (GI) tract. It is estimated to affect 3 million people in the US with the incidence continuing to rise.1 Unfortunately, IBD has a complex etiology2,3 and a significant number of patients do not benefit from current standard-of-care treatments.4,5 Anti-TNF therapy is commonly used to manage symptoms; however, primary non-response rates range from 10-30%, with up to 46% of patients experiencing a symptomatic recurrence after one year.6,7
During the later stages of preclinical drug development, lead compounds are tested in animals to determine if they are safe and effective. In most countries, pharmacologists are legally required to test new drugs in animals before clinical trials. Species differences can however mean that animal data doesn't translate accurately to humans. For example, many mouse IBD models display differences in protein expression from humans that can influence drug behavior.
Human tissue models for IBD Research
To improve confidence heading into clinical trials, researchers may decide to support their animal studies with data from human fresh ex vivo tissue models, or 3D bioengineered tissue models, to:
- avoid species differences which may affect drug safety or efficacy data
- link human drug response to genetic factors e.g., biomarkers that indicate efficacy
- ensure that animal data matches what is observed in humans before clinical trials
- give mechanistic insights into why a drug response may differ between humans and animals
An additional benefit of using fresh human tissue for IBD research is that patient drug treatment history can be correlated to drug response. The combined use of biomarker and mRNA analysis can identify genes with the potential to predict drug performance, or whether that patient will be a ‘responder' or ‘non-responder'. This can eliminate the need to recruit entire patient populations and allows drug developers to aim trials at a specific patient cohort.
References
- Centre for Disease Control and Prevention (CDC). Inflammatory Bowel Disease (IBD): Prevalance of IBD (2022).
- Burish. Inflammatory bowel disease epidemiology. Current Opinion Gastroenterology 29:4 (2013).
- Steenholdt et al. Optimizing treatment with TNF inhibitors in inflammatory bowel disease by monitoring drug levels and antidrug antibodies. Inflammatory Bowel Diseases 22:8 (2016).
- Porter et al. Can we target endogenous anti-inflammatory responses as a therapeutic strategy for inflammatory bowel disease? Inflammatory Bowel Disease (2018).
- Sanger. Gastrointestinal pharmacology: challenges ahead. Frontiers in Pharmacology 1 (2010).
- Ananthakrishnan. Epidemiology and risk factors for IBD. Nature Reviews Gastroenterology and Hepatology 12 (2015).
- Schork. Time for one-person trials. Nature 520 (2015).
.jpg?width=756&height=425&name=New%20Approach%20Methodologies%20(NAMs).jpg)

