Case Study
From Clinical iPSC Seed Clone to
U.S. Phase 3 Cell Therapy
REPROCELL’s StemRNA™ Clinical Seed Clone VCT-37-F35 Supports a Phase 3 Fertility Therapy
Gameto’s Fertilo program uses iPSC-derived ovarian support cells to mature eggs outside the body as part of their next-generation IVF technology.
REPROCELL’s StemRNA™ Clinical Seed Clone VCT-37-F35 (Female, 23, Caucasian, Healthy, A+) served as the starting iPSC source for the program.

High-Quality iPSCs: The Foundation for Clinical Therapy
Developing an iPSC-based clinical therapy requires starting cells that support:
• Clinical manufacturing workflows
• Reproducible differentiation into therapeutic cell types
• Regulatory documentation suitable for clinical programs
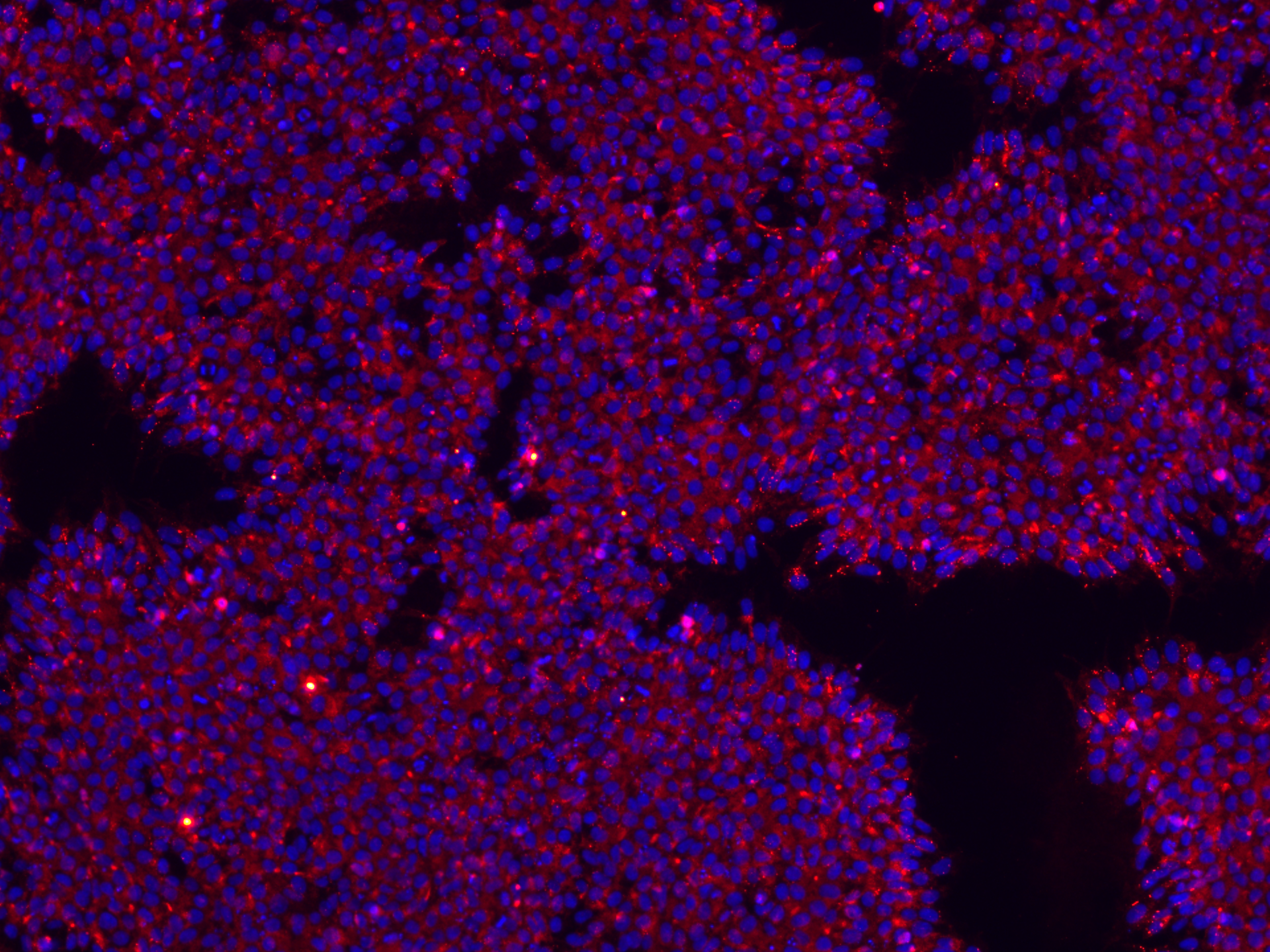
For Fertilo, the goal was to generate ovarian support cells (OSCs) from iPSCs that could replicate the natural ovarian environment and support egg maturation outside the body.

Key elements of the workflow:
• REPROCELL's seed clone was generated using non-integrative mRNA reprogramming technology
• Manufactured following GMP principles at REPROCELL's Japan GMP facility, and supplied via REPROCELL USA.
• Used to generate clinical seed banks and master cell banks
• Differentiated into ovarian support cells (OSC) for Fertilo development
This approach provides a clinically qualified starting point for cell therapy development.

From First Live Birth to FDA-Cleared Phase 3 Trial
The in vitro oocyte maturation technology helped enable the world’s first live birth using Fertilo, demonstrating the clinical potential of iPSC-derived cells in reproductive medicine.
Fertilo received FDA IND clearance, enabling the first Phase 3 clinical trial in the U.S. for an iPSC-based therapy.
Why This Matters for Cell Therapy Developers?
Proven iPSC Quality for Clinical Translation
REPROCELL’s involvement in the Fertilo program highlights how high-quality clinical iPSC starting material can support real clinical innovation. StemRNA™ Clinical iPSC Seed Clones are designed to provide a reliable, regulatory-aligned foundation for advanced cell therapy development, combining
GMP-aligned manufacturing, robust characterization, and documented use in clinical programs.
- Clinically validated starting material – StemRNA™ Clinical iPSC Seed Clones are manufactured under GMP principles and have supported real clinical programs, including a U.S. Phase 3 iPSC-based therapy.
- Consistent differentiation performance – Clonal, well-characterized iPSC lines enable reproducible differentiation and reliable downstream cell therapy product manufacturing.
- Strong quality & genetic integrity controls – Comprehensive characterization includes donor screening, karyotyping, whole genome oncogenic analysis, and pluripotency validation.
- Built for regulatory pathways – Our Clinical iPSC seed clones are supported by regulatory documentation, including FDA Drug Master Files, helping streamline IND filings and clinical development.
References:
-
Bruna Paulsen et al. Development of human induced pluripotent stem cell-derived ovarian support cells as a clinical-grade product for in vitro fertilization. Cell Stem Cell, 33, 1-15 (5 Feb 2026).
DOI: 10.1016/j.stem.2025.12.020
Explore Relevant Pages:
Contact our experts
At REPROCELL, our scientists understand that your custom iPSC project must be as unique as your research. If you have any questions about how our Clinical iPSC Generation Service can help you advance your project, please make an inquiry using the form below.
Discover More
Resources
- FAQ: Clinical iPSCs
- FAQ: Clinical MSCs
- FAQ: iPSC-Derived Exosomes
- Making iPSC-Derived Therapeutics a Clinical Reality – our external article in the European Biopharmaceutical Review.
Gene Editing Services
Latest in Stem Cells

What Are Off-Target Effects in Cellular Gene Editing? Risks, Detection, and Solutions
Explore off-target effects in gene editing, from CRISPR risks and AI-driven detection to genomic safety in iPSC therapies.
20 May 2026

5 Hard Truths About Bringing Stem Cell Therapies to the Clinic
Discover the essential truths behind developing clinical-grade stem cell therapies and learn how to navigate the complexities of GMP manufacturing successfully.
30 March 2026

REPROCELL Attends Japan-Maryland Night 2026
REPROCELL was honored to attend Japan-Maryland Night on Wednesday March 4th 2026 Maryland State, USA, hosted by the Embassy of Japan (in Washington DC).
06 March 2026