Ready-to-use StemEdit Hypoimmune Lines:
What’s New
Optimized for Reduced Immunogenicity and Allogeneic Compatibility
REPROCELL’s StemEdit Hypoimmune iPSC lines will be immediately available, can be evaluated under research conditions, and allow you to easily progress to clinical translation. Built on one of our StemRNA™ Clinical iPSC Seed Clones, these lines are generated using our proprietary genome editing system and reprogrammed with footprint-free StemRNA™ technology under GMP principles.
Hypoimmune StemEdit Lines
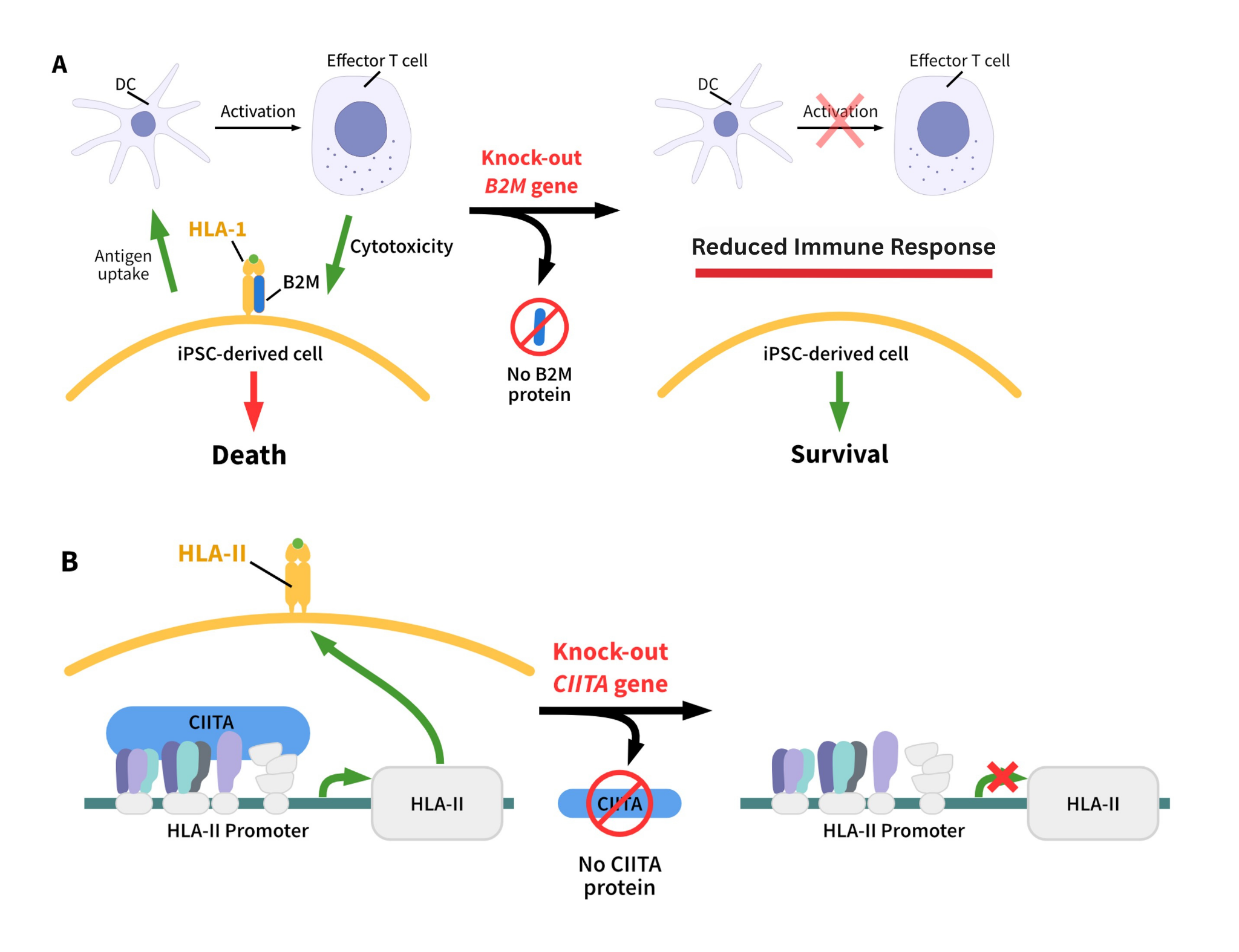
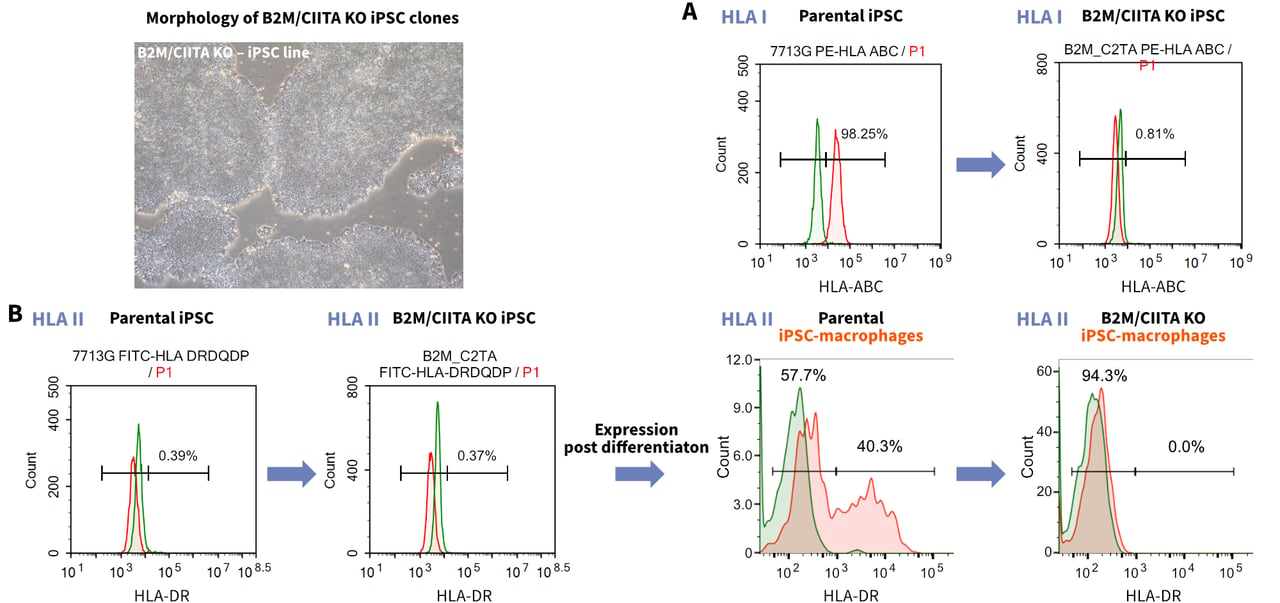
- HLA Class I/II Double Knockout
StemEdit Human iPSC non-HLA Class I/II (B2M/CIITA Homo Double KO) - HLA Class I Knockout
StemEdit Human iPSC non-HLA Class I (B2M Homo KO) - HLA Class II Knockout
StemEdit Human iPSC non-HLA Class II (CIITA Homo KO)
We also offer GMP iPSC Master Cell Bank (MCB) vials derived from the parental iPSC line as an affordable starting point for your own clinical gene editing projects and custom Hypoimmune line generation.
For detailed specifications, pricing, or to discuss your project needs, contact us at info-us@reprocell.com.
Key Benefits
- Standardized, well-characterized genetic modifications
- Immediate availability for research programs
- Clear pathway to matched clinical-grade seed clones
- GMP-aligned development and traceability
- Ready for both research use and clinical translation
Our off-the-shelf lines provide speed for your research program without closing the door on future clinical development.