StemRNA™ Clinical iPSCs
Quality Control & Characterization Data
Human iPSCs can differentiate into a wide range of specialized cell types, enabling applications in regenerative medicine. REPROCELL’s StemRNA™ Clinical iPSC Seed Clones are generated using integration-free RNA reprogramming and undergo extensive quality control to ensure genetic integrity and suitability for downstream clinical manufacturing.
StemRNA™ Clinical iPSC Clones
Functional Differentiation into Cytotoxic iNK Cells
Live-cell imaging of human iNK cells interacting with tumor K562 cells. Elongated human iNK (induced Natural Killer) cells exhibit cytotoxic activity when co-cultured with CFSE-labeled K562 cells (green, round), a human chronic myeloid leukemia cell line. These data demonstrate that iNK cells display mature functional capacity to recognize and eliminate target cells.
Data were generated in collaboration with the Tokyo Metropolitan Institute of Medical Science, Japan.
Every StemRNA Clinical iPSC Seed Clone, along with its donor material and any subsequent GMP MCB batch undergoes comprehensive genetic integrity testing. Low-resolution G-band karyotyping confirms normal overall chromosome structure and the absence of numeric aberrations and structural aberrations. For higher resolution analysis, whole genome sequencing-based oncogenic screening evaluates genetic variants across more than 400 cancer-related genes. A detailed oncogenic report which analyses interprets high-impact genetic variants using publicly available clinical databases, is provided for each ready-to-use clinical seed clone.
Quality Control Data of Typical StemRNA™ Clinical iPSC Clones
Morphology, P6+3

Karyology

Flow cytometry analysis

3-germ layers differentiation potential measured by RT qPCR

Typical quality control assays of Clinical StemRNA iPSC Lines. Typical quality control assays for Clinical StemRNA™ iPSC lines. To confirm suitability as starting material for clinical programs, each clonal line undergoes comprehensive validation, including cell count, post-thaw viability, sterility and mycoplasma testing, colony morphology and growth assessment, karyotyping, and pluripotency marker expression by flow cytometry. Functional pluripotency is further evaluated through directed differentiation into the three germ layers. Only clones meeting all batch release specifications are advanced to scale-up for GMP Master Cell Bank manufacturing. Data generated in-house.
Efficient Neural Differentiation of StemRNA™ iPSCs
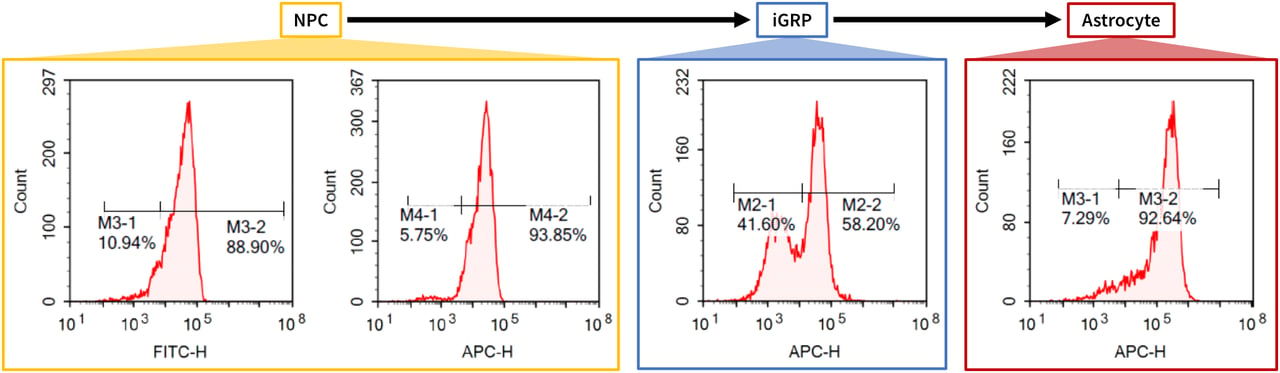
Typical targeted differentiation of StemRNA™ clinical iPSC clones into the neural lineage. Cells were sequentially differentiated and assessed by flow cytometry, showing expression of stage-specific markers for neural progenitor cells, iGRPs (induced glia-restricted progenitor cells), and astrocytes. Data generated in-house.
Typical iNK Cell Differentiation of
StemRNA™ Clinical iPSC Clones

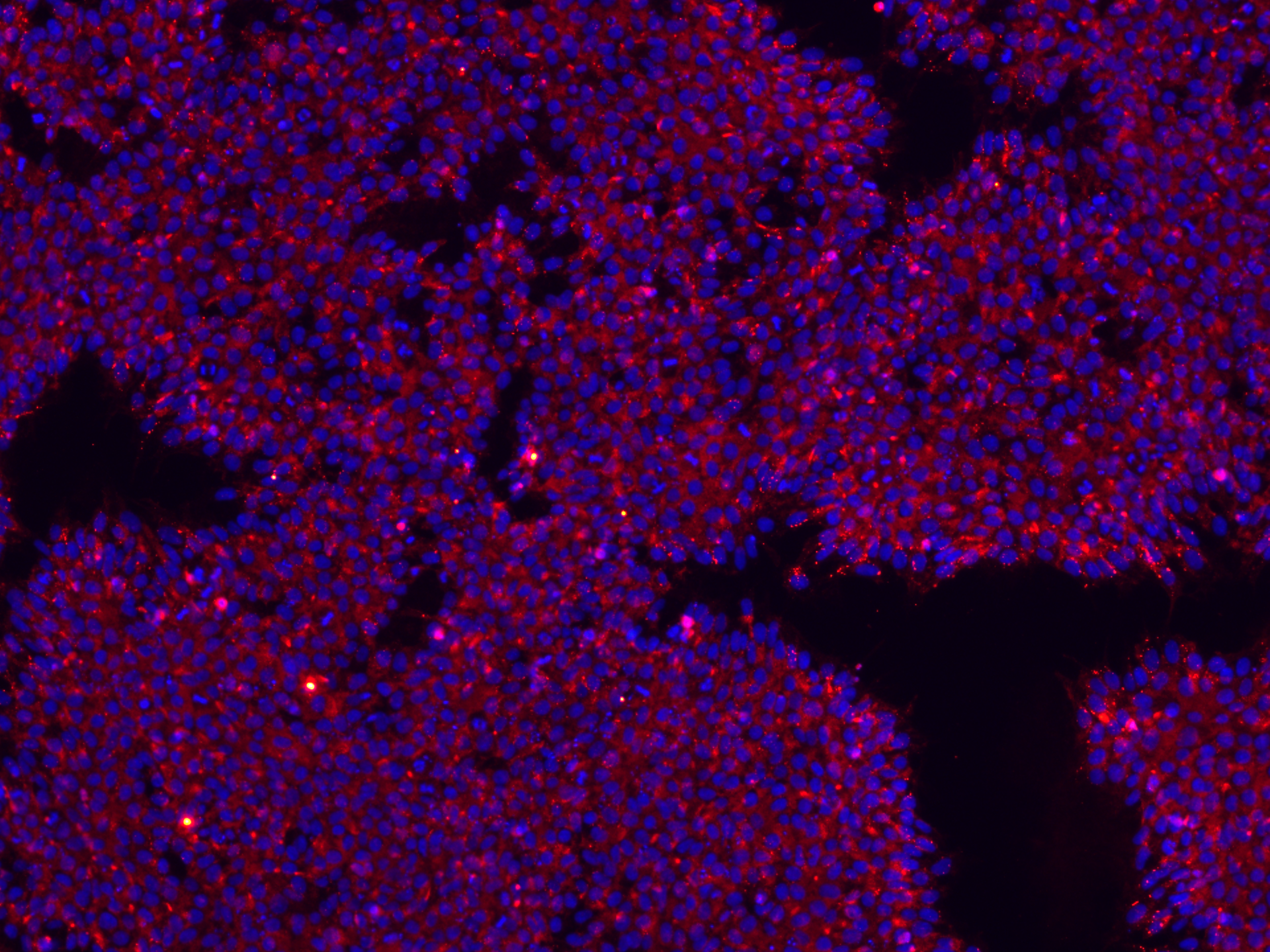
Expression of key NK cell surface markers in iNK cells derived from StemRNA™ clinical iPSC clones REPROCELL's clinical iPSCs were differentiated into iNK cells and characterized by flow cytometry. CD45⁺CD56⁺ NK cells expressed NKp44 (CD336), NKp46 (CD335), and NKG2D (CD314), demonstrating markers typical of mature, functional iNK cells.
Data generated in collaboration with the Tokyo Metropolitan Institute of Medical Science, Japan.
Contact our experts
At REPROCELL, our scientists understand that your custom iPSC project must be as unique as your research. If you have any questions about how our Clinical iPSC Generation Service can help you advance your project, please make an inquiry using the form below.
Discover More
Resources
- FAQ: Clinical iPSCs
- FAQ: Clinical MSCs
- FAQ: iPSC-Derived Exosomes
- Making iPSC-Derived Therapeutics a Clinical Reality – our external article in the European Biopharmaceutical Review.
Gene Editing Services
Latest in Stem Cells

What Are Off-Target Effects in Cellular Gene Editing? Risks, Detection, and Solutions
Explore off-target effects in gene editing, from CRISPR risks and AI-driven detection to genomic safety in iPSC therapies.
20 May 2026

5 Hard Truths About Bringing Stem Cell Therapies to the Clinic
Discover the essential truths behind developing clinical-grade stem cell therapies and learn how to navigate the complexities of GMP manufacturing successfully.
30 March 2026

REPROCELL Attends Japan-Maryland Night 2026
REPROCELL was honored to attend Japan-Maryland Night on Wednesday March 4th 2026 Maryland State, USA, hosted by the Embassy of Japan (in Washington DC).
06 March 2026