Example Protocol for the Culture of the Lung Carcinoma A549 Cell Line on Alvetex® Scaffold in Well Insert and Well Plate Formats
● Download this protocol as a PDF (1.6 MB)
1. Introduction
A549 is a human alveolar basal epithelial cell line isolated in the early 1970s from lung cancerous tissue[1]. A549 cells retain characteristics of type II alveolar epithelium in vitro, such as production of lecithin[2], expression of cytochrome P450 enzymes[3] and presence of lamellar bodies[3]. They are also extensively used in cytotoxicity models of non-small-cell lung cancer, and have been shown to be responsive to tamoxifen, in accordance with their expression of oestrogen receptors and binding sites[4], as well as cisplatin[5].
Alvetex Scaffold is available in several cell culture formats including 24 well plate (AVP006), 12 well plate (AVP002), 6 well insert (AVP004), 12 well insert (AVP005), and 24 well insert (AVP012).
24 well and 12 well plates are suitable for shorter term cultures and for applications where limited cell penetration into the scaffold is required. Well insert formats generally support longer term cultures and deeper cell penetration into the scaffold. They also provide for conveniently tailored media set ups (see the Alvetex Scaffold Quick Start protocol).
The availability of two different well insert formats enables choice on the basis of desired culture size and cell expenditure. 6 well inserts can be placed in conventional 6 well plates, while 12 well inserts can be placed in either 6 well plates or 12 well plates, depending on media requirements. Alternatively, both insert types can be housed in the dedicated Well Insert Holder in Deep Well Petri Dish (AVP015) to allow for increased media volumes and prolonged cell culture.
2. Methods
2.1. Preparation for 3D Cell Culture on Alvetex Scaffold
- A549 cells (ATCC, CCL-185) were routinely maintained in T-75 flasks.
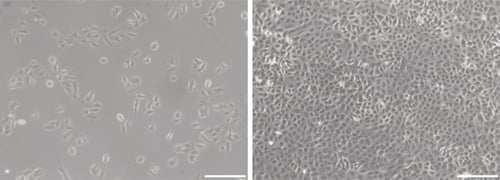
Figure 1. Phase contrast micrographs of 3T3 cells grown in conventional 2D culture plates. Images show cells at low (left) and high (right) confluency. Scale bars: 200 μm.
- Complete growth media consisted of: F-12K nutrient mixture (Kaighn’s modification) supplemented with 10 % v/v FBS and 100 U/ml Penicillin/Streptomycin.
Note: F-12K medium already contains 2 9M L-glutamine.
- Cells were harvested by trypsinisation and centrifuged for 5 minutes (1000 rpm). The supernatant was discarded and the cell pellet was re-suspended in an appropriate volume of media for cell counting by Trypan Blue.
Note: F-12K medium already contains 29M L-glutamine.
- Cells were re-suspended at a concentration of 1.0 × 107 cells/ml for seeding.
2.2. 24 well Plate Format (AVP006)
-
Alvetex Scaffold 24 well plates were prepared for seeding with a 70 % ethanol wash (2 mL per well) and subsequent media washes (twice with 2 ml of media each).
-
25 μl of the cell suspension was added to the centre of the Alvetex Scaffold disc, which was equivalent to 0.25 × 106 cells per well.
-
The plate was incubated for at least 1 hour at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
-
2 mL of media was added to each well taking care not to dislodge cells from the Alvetex Scaffold.
-
Plates were re-incubated and maintained by complete media exchange every other day.
Note: This method can be applied to the use of Alvetex Scaffold in 12 well plate format (AVP002). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start protocol.
2.3. 12 well Insert Format (AVP005)
- Alvetex Scaffold 12 well inserts in 12 well plate format were prepared for seeding by dipping in 70 % ethanol followed by media washes (twice with 4 mL per well).
- Wells were filled from the outside of the insert with enough medium to allow it to rise inside the insert and cover the substrate, but not to go over the sides of the inserts, i.e. 2.4 mL ± 0.2 mL, as described for feeding above and below separately in the Quick Start Protocol.
- 25 μL of the cell suspension was added in droplets over the surface of the Alvetex Scaffold disc, which was equivalent to 0.25 × 106 cells per well.
- The plate was incubated overnight at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
- Media was added to each well to a total volume of 4 mL the following morning taking care not to dislodge cells from the Alvetex Scaffold.
- Plates were re-incubated and maintained by complete media exchange every other day.
Note: This method can be applied to the use of Alvetex Scaffold in 6 well insert format (AVP004). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start protocol.
3. Example Data
3.1. 24 well plate format (AVP006)
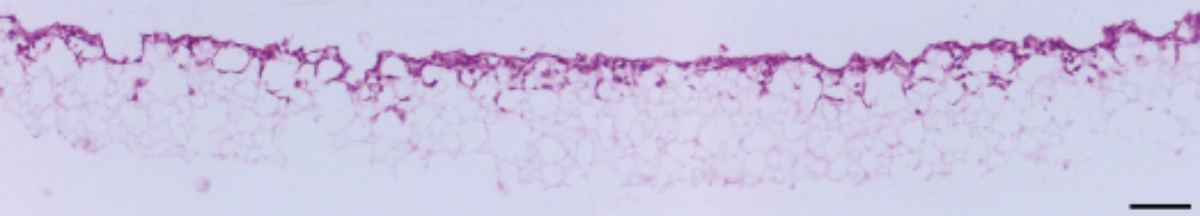
Figure 2. Brightfield micrograph showing the structure of A549 cells cultured for 7 days on 15 mm diameter Alvetex Scaffold discs presented in 24 well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 µm) and counterstained with haematoxylin and eosin. Scale bar: 100 µm.
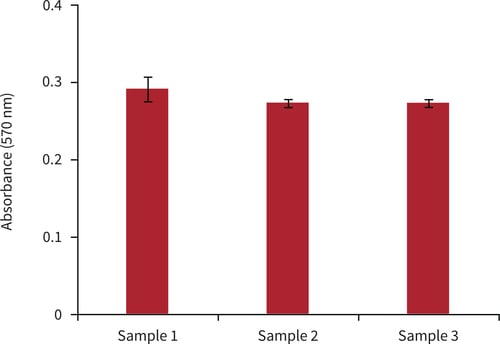
Figure 3. Biochemical analysis of cell viability using a standard MTT assay. Data from 3 sample replicates of A549 cells are shown, each sampled in triplicate (n = 3, mean ± SD). Cells were cultured for 3 days on 15 mm Alvetex Scaffold discs presented in 24 well plate format.
3.2. 12 well insert format (AVP005)
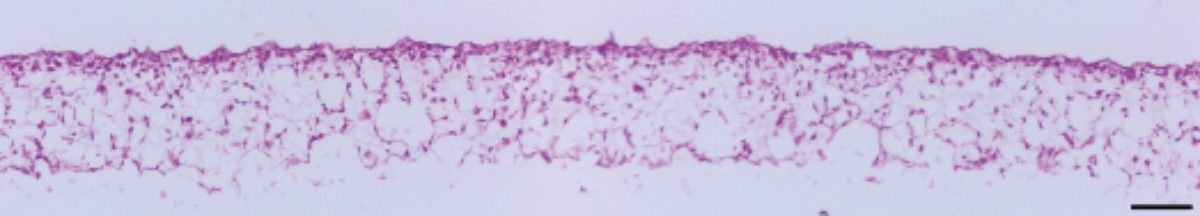
Figure 4. Brightfield micrograph showing the structure of A549 cells cultured for 7 days on 15 mm diameter Alvetex Scaffold discs presented in 12 well insert in 12 well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 µm) and counterstained with haematoxylin and eosin. Scale bar: 100 µm.
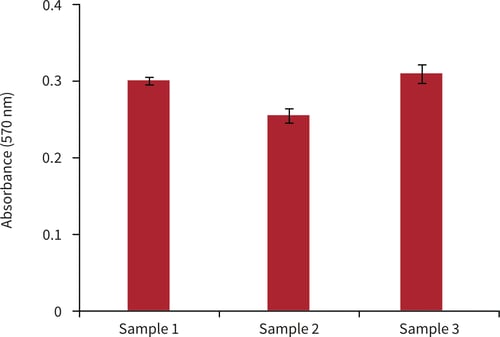
Figure 5. Biochemical analysis of cell viability using a standard MTT assay. Data from 3 sample replicates of A549 cells are shown, each sampled in triplicates (n = 3, mean ± SD). Cells were cultured for 3 days on 15 mm Alvetex Scaffold discs presented in 12 well inserts in 12 well plate format.
4. References
-
Giard DJ et al, 1973. In vitro cultivation of human tumors: establishment of cell lines derived from a series of solid tumors. J Natl Cancer Inst 51(5):1417-23.
-
Lieber M et al, 1976. A continuous tumor-cell line from a human lung carcinoma with properties of type II alveolar epithelial cells. Int J Cancer 17(1):62-70.
-
Foster KA et al, 1998. Characterization of the A549 cell line as a type II pulmonary epithelial cell model for drug metabolism. Exp Cell Res 243(2):359-66.
-
Caltagirone S et al, 1997. Interaction with type II estrogen binding sites and anti- proliferative activity of tamoxifen and quercetin in human non-small-cell lung cancer. Am J Respir Cell Mol Biol 17(1):51-9.
-
Chen J et al, 2010. Resistance to platinum-based chemotherapy in lung cancer cell lines. Cancer Chemother Pharmacol 66(6):1103-11.