Clinical-grade iPSCs form the foundation of allogeneic cell therapy programs. Choosing the right starting cell line shapes regulatory strategy, streamlines manufacturing timelines, and sets the stage for long-term commercialization, making early decisions critical because they are often difficult or impossible to reverse later.
REPROCELL’s StemRNA™ Clinical iPSC Seed Clones are designed to provide an FDA, EMA, and PMDA, regulatory-aligned starting point, supporting translational stem cell therapy development. These cells are generated using REPROCELL’s proprietary non-integrative, mRNA-based reprogramming technology (Figure 1). They are manufactured at REPROCELL Japan and REPROCELL USA using manufacturing processes that follow GMP principles and use clinical-grade reagents to ensure high quality and consistency.
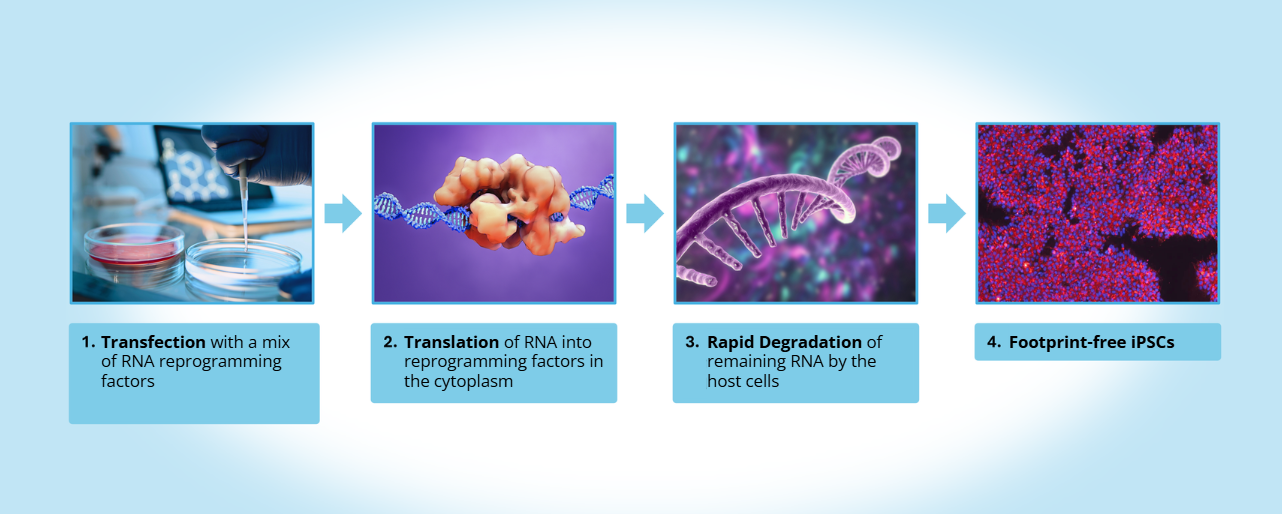
Figure 1. mRNA-based reprogramming enables generation of footprint-free iPSCs. Somatic cells are transfected with a synthetic mRNA cocktail encoding a proprietary set of reprogramming factors. The mRNAs are translated in the cytoplasm to produce reprogramming proteins and are rapidly degraded by the host cell. As no DNA intermediates or vector integration are involved, this approach generates clinically suitable, integration-free iPSCs with no detectable genomic footprint.
Top 5 Advantages of StemRNA Clinical iPSC Seed Clones
1. Seed Clone Used in a U.S. Clinical Phase 3 iPSC Therapy Program
One of our iPSC Seed Clones - VCT-37-F35, supports Gameto’s Fertilo in vitro oocyte maturation technology that shortens standard IVF treatment. This clone served as the starting material for a master cell bank (MBC). This MCB was then used for differentiation into ovarian support cells, which are now part of the first U.S. Phase 3 iPSC-based therapy trial.
| Program impact | Clinical seed clone with demonstrated use in an FDA U.S. Phase 3 iPSC therapy program. |
2. Active Drug Master File (DMF)
All U.S.-manufactured StemRNA™ Clinical Seed Clones are supported by an active FDA DMF, accessible through a Letter of Authorization.
| Program impact | Simplified regulatory submissions and reduced burden of generating foundational documentation. |
3. Documented FDA engagement
REPROCELL’s iPSC Seed platform has supported multiple INTERACT meetings and IND submissions addressing key topics such as donor eligibility, cell line characterization, manufacturing controls, genetic stability, and release testing. Developers benefit from a platform already reviewed in clinical contexts, with supporting documentation aligned to FDA expectations.
| Program impact | This prior regulatory experience supports a more predictable path from seed clone selection to IND-enabled cell manufacturing. |
4. Oncogenic Analysis Across Seed Clone, Donor Material & any GMP MCB batches
Each seed clone, along with its donor material and any subsequent GMP MCB batch undergoes comprehensive genetic integrity testing. Low-resolution G-band karyotyping confirms normal overall chromosome structure and the absence of numeric aberrations and structural aberrations. For higher resolution analysis, whole genome sequencing-based oncogenic screening evaluates genetic variants across more than 400 cancer-related genes. A detailed oncogenic report which analyses interprets high-impact genetic variants using publicly available clinical databases, is provided for each ready-to-use clinical seed clone.
| Program impact | Strong genetic integrity data for regulatory submissions. |
5. Straightforward Commercial and Licensing Structure
All donor tissues used to generate StemRNA™ Clinical iPSC Seed Clones are fully consented for both clinical and commercial use, eliminating uncertainties around donor rights and downstream product commercialization. Developers can secure the necessary rights early in the program.
| Program impact | Reduces legal complexity, avoids renegotiation later, and supports a smoother transition into clinical and commercial manufacturing. |
Foundation for GMP MCB Expansion and StemEdit Gene Editing
StemRNA™ Clinical iPSC Seed Clones provide a ready starting point for GMP MCB manufacturing. There are three main options for scaling up the GMP-like seed clones (Figure 2):
1. Gene Editing with StemEdit – Targeted edits are introduced at the seed stage before GMP MCB expansion, enabling gene-edited GMP iPSC.
2. Direct GMP Expansion – Unedited StemRNA™ Clinical iPSC Seed Clones can be scaled to GMP iPSC MCB and working cell banks (WCBs).
3. Direct GMP iMSC Production – Seed clones can be differentiated and expanded directly into GMP iPSC-derived mesenchymal stem cell (iMSC) MCBs. iMSCs are widely used in regenerative medicine due to their immunomodulatory properties and scalability. Their origin from a clonal iPSC source enables consistent, standardized allogeneic cell therapy manufacturing.
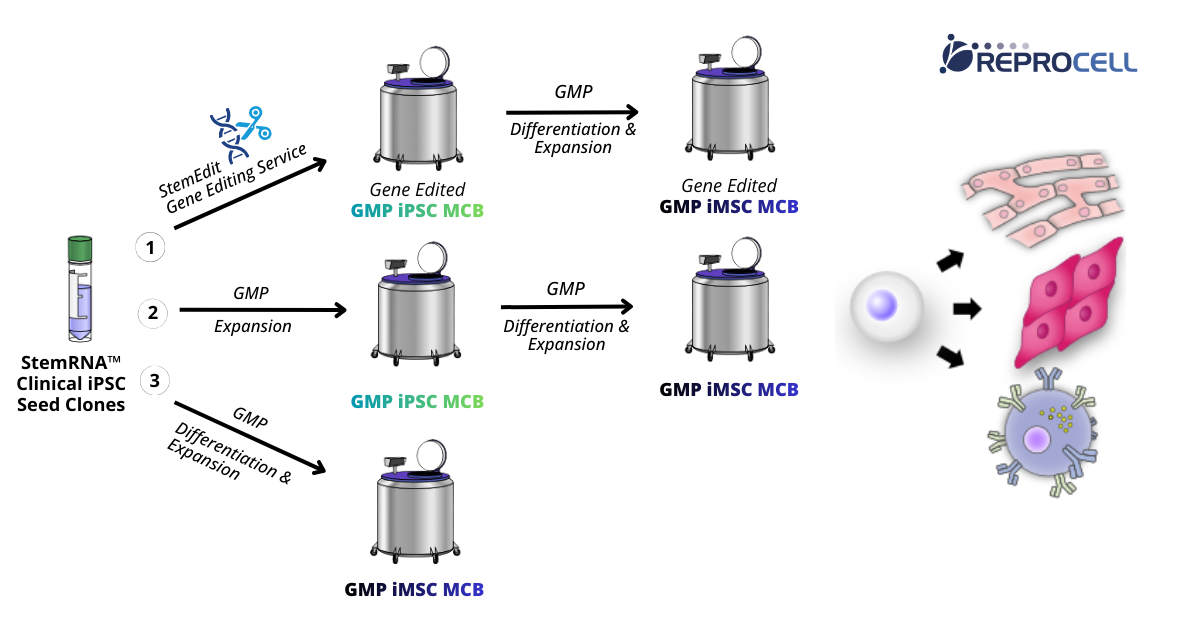
Figure 2. StemRNA™ Clinical iPSC Seed Clones serve as a starting material for GMP iPSC and iMSC MCB generation. Seed clones can be either genetically modified prior to expansion and MCB generation, expanded directly into a GMP iPSC banks, or first differentiated in GMP and scaled into a GMP iMSC bank.
Corresponding StemRNA™ Clinical Pilot Clones are also available as research-use counterparts to each Clinical Seed Clone. Because Pilot Clones originate from the same clonal source, they allow developers to optimize differentiation protocols, perform proof-of-concept studies, and refine manufacturing workflows while preserving the Clinical Seed Clone for direct GMP Master Cell Bank generation. Both seed and pilot clones include an evaluation period to determine suitability for the developer’s process. During this period, sufficient vials of the selected seed clone are reserved, enabling direct progression into GMP iPSC MCB generation after successful evaluation.
| Program impact | Direct path from seed clone to gene edited or unedited GMP iPSC and iMSC MCBs without restarting clone selection. |
Frequently asked questions about StemRNA™ clinical iPSC seed clones
-
What is a clinical iPSC seed clone?
A clinical iPSC seed clone is a fully characterized, consented, and quality-controlled induced pluripotent stem cell line used as the starting material for GMP master cell bank manufacturing.
-
Why use a seed clone instead of reprogramming your own iPSC line?
Using a pre-qualified seed clone with an active DMF and regulatory history can reduce development time, simplify IND filings, and lower regulatory risk.
-
Can clinical iPSC seed clones be gene edited?
Yes. With StemEdit services, seed stock clones can be edited prior to GMP MCB expansion, allowing developers to introduce therapeutic edits while maintaining a clear regulatory path.
-
Are StemRNA seed clones used in clinical trials?
Yes. A StemRNA™ Clinical iPSC Seed Clone supported the development of the Fertilo program which has advanced into Phase 3 clinical programs in the United States.
-
Do StemRNA seed clones support commercial manufacturing?
Yes. All donor tissues are fully consented for clinical and commercial use, and clear licensing options are available to support long-term product commercialization.
Relevant Webpages
Explore StemRNA™ Clinical iPSC Seed Clones and learn how they can accelerate your path to GMP MCB and iMSC manufacturing.
-
StemRNA Clinical iPSC Seed Clones: Learn more about StemRNA Clinical iPSC Seed Clones
-
Gameto Fertilo Technical Summary: https://reprocell.com/hubfs/StemRNAClinicalSeedCloneFertiloTechnicalSummary.pdf
-
GMP‑Grade iMSC & MSC Manufacturing & MCB Generation: https://www.reprocell.com/clinical-stem-cell-services/gmp-imsc-and-msc-production
-
Clinical-grade iPSCs and GMP MCBs FAQ: https://www.reprocell.com/clinical-stem-cell-services/faq-ipscs
Contact REPROCELL
To discuss your GMP iPSC or iMSC manufacturing projects, contact our team at info-us@reprocell.com.


.png?width=1000&height=562&name=hubspot_featured_image_1200x628%20(1).png)

%20(1).png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs)%20(1).png)
.jpg?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).jpg)

