MTS Cell Viability Assay of upcyte® Hepatocyte Cultures Grown on Alvetex® Scaffold in 3D
● Download this protocol as a PDF (0.3 MB)
1. Introduction
This chromogenic assay involves the biological reduction by viable cells of the tetrazolium compound 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H- tetrazolium (or MTS). The MTS assay reagent is composed of MTS and the electron coupling agent phenazine methosulfate (PMS). The formazan product of MTS reduction is soluble in tissue culture medium. This reaction only takes place when mitochondrial reductase enzymes are active, and therefore the conversion can be directly related to the viability of cells in culture. The MTS reagent alone results in very low background absorbance values in the absence of cells. It is recommended that for each cell type the linear relationship between cell number and signal produced should be first established in order to investigate the limits of the assay.
2. Methods
These are example protocols which may require further optimisation depending on the rate of cell growth of a particular cell type on Alvetex Scaffold. The protocols describe cell viability analysis of upcyte® hepatocytes (upcyte® Technologies GmBH) using the CellTiter 96® Aqueous Non-Radioactive Cell Proliferation Assay (Promega). Upcyte® cells were thawed, immediately seeded onto the Alvetex Scaffolds and medium added after a 3 hour attachment period. At various timepoints, the growth and viability of the upcyte® hepatocytes were monitored as described below.
2.1. Method 1: Performing the MTS assay on dismantled Alvetex Scaffold discs
- Prepare sufficient MTS/PMS solution for the experiment, including controls, by mixing the PMS solution with the MTS solution according to the manufacturer’s instructions (Promega, G5421). Briefly this involves mixing 1 mL PMS solution per 20 mL MTS solution. The resulting mixture can be aliquoted and stored at –20 °C before use.
- Dilute the MTS/PMS solution 1:5 in appropriate diluent (e.g. PBS, or phenol red-free culture medium).
- Remove the Alvetex Scaffold disc from the culture vessel and dismantle inserts where applicable. For 22 mm Alvetex Scaffold discs, transfer to the wells of a clean 12 well plate. For 15 mm Alvetex Scaffold discs, transfer to the wells of a clean 24 well plate.
- For 22 mm Alvetex Scaffold discs add 0.8 mL of diluted MTS/PMS solution to each well. Also add 0.8 mL diluted MTS/PMS solution to empty control wells containing no cells. For 15 mm Alvetex Scaffold discs add 0.4 mL of diluted MTS/PMS solution to each well. Also add 0.4 mL diluted MTS/PMS solution to empty control wells containing no cells.
- Incubate samples at 37 °C and 5 % CO2 for 1 hour.
- Transfer 100 µL aliquots of each sample to replicate wells of a clear 96 well plate.
- Measure the absorbance of the samples and controls at 490 nm.
2.2. Method 2: Performing the MTS assay on Alvetex Scaffold in situ in well inserts
- Prepare sufficient MTS/PMS solution for the experiment, including controls, by mixing the PMS solution with the MTS solution according to the manufacturer’s instructions (Promega, G5421). Briefly this involves mixing 1 mL PMS solution per 20 mL MTS solution. The resulting mixture can be aliquoted and stored at –20 °C before use.
- Dilute the MTS/PMS solution 1:5 in appropriate diluent (e.g. PBS, or phenol red-free culture medium).
- Remove the Alvetex Scaffold insert from the culture vessel and transfer to a clean plate. For 22 mm Alvetex Scaffold inserts, transfer to a clean 6 well plate. For 15 mm Alvetex Scaffold inserts, transfer to a clean 12 well plate.
- For 22 mm Alvetex Scaffold inserts add 7 mL of diluted MTS/PMS solution to each well. For 15 mm Alvetex Scaffold inserts add 3 mL of diluted MTS/PMS solution to each well. Add the same corresponding volume of diluted MTS/PMS solution to empty control wells containing no cells.
- Incubate samples at 37 °C and 5 % CO2 for 3 hours.
- Transfer 100 µL aliquots of each sample to replicate wells of a clear 96 well plate.
- Measure the absorbance of the samples and controls at 490 nm.
3. Example Data Set
The example data shown below was obtained from cutures of upcyte® hepatocytes grown in 15 mm Alvetex Scaffold presented in 12 well insert format.
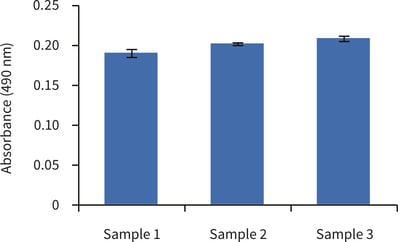
Figure 1. Biochemical analysis of cell viability using a standard MTS assay according to Method 2, above. Data from 3 sample replicates of upcyte® hepatocytes cultured in Alvetex Scaffold in 12 well insert format are shown (n = 3, mean ± SD). Note good reproducibility between samples.
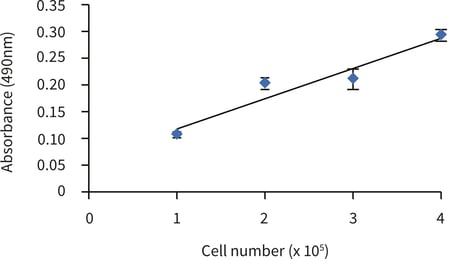
Figure 2. MTS assay of upcyte® hepatocytes cultured for 5 hours in Alvetex Scaffold discs presented in 12 well insert format (n = 3, mean ± SD). Good linearity was observed (R2 > 0.9) between cell number and signal produced when cells were seeded at 1.0, 2.0, 3.0 and 4.0 × 105 cells per disc.
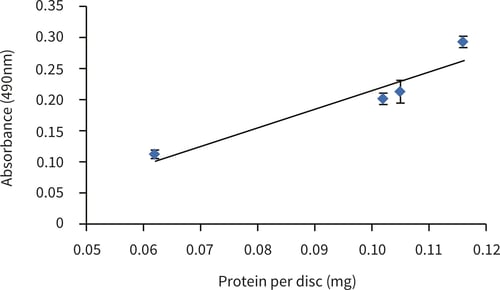
Figure 3. MTS assay of upcyte® hepatocytes cultured for 5 hours in Alvetex Scaffold discs presented in 12 well insert format (n = 3, mean ± SD). Data normalised to protein extracted from Alvetex Scaffold discs seeded at 1.0, 2.0, 3.0 and 4.0 × 105 cells. Good linearity was observed (R2 = 0.9) between cellular protein and MTS viability reading.