Gastrointestinal metabolism assay
Drug Discovery Assay – reference number: B116
| Assay type: | Gastrointestinal (GI) tract |
| Tissue: | Human colon/ human gastrointestinal mucosa (healthy) |
| Target: | Phase1/ Phase 2 metabolic enzymes |
| Control compound: | Phase1/ Phase 2 metabolic enzymes |
| Study type: | Ussing chamber |
| Functional endpoint: | Metabolism |
Assay Description
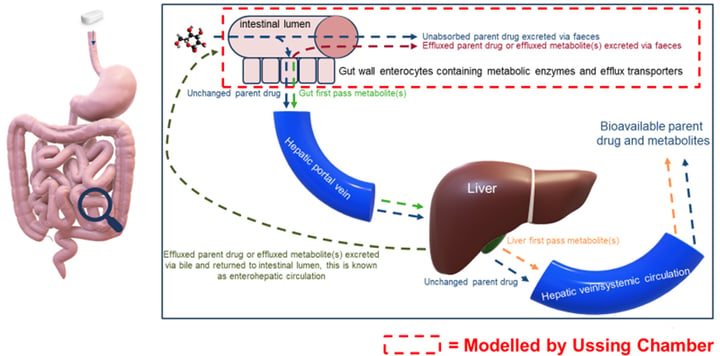
This assay assesses the metabolism of test articles across human gastrointestinal mucosa. The intestines are essential for the digestion of food and the absorption of fluid and electrolytes. Changes in the anatomy or function of the intestines can lead to disorders such as Irritable Bowel Syndrome (IBS), Crohn’s, Ulcerative Colitis, and diarrhea.
Testing Information
Introduction
The specific results that will be provided are on the metabolism of test articles across human gastrointestinal mucosa.
Test Article Requirements
Test article(s) to be provided by the Sponsor in storable aliquots at required test concentrations with information on diluent vehicle used. Stock solutions are prepared in deionized water unless otherwise requested. Bath volumes are 5mL; sponsor to provide sufficient test article to run the entire study.
Suggested Testing
We recommend testing in duplicate replicates per donor.
Study Outline
Rationale and Experimental Design
This assay assesses the metabolism of test articles across human gastrointestinal mucosa.
Exclusion Criteria
No specific exclusion criteria are in place other than to reject macroscopically diseased/necrotic tissue. Furthermore, tissues which do not have a satisfactory potential difference or transepithelial resistance will be excluded.
Standardization and Qualification
All GI mucosal tissues are initially processed through standardization and qualification procedures to ensure functionality, prior to starting the study protocol.
Prior to mounting the mucosa in the Ussing Chambers, the potential difference of the electrodes and the resistance due to the PSS will be offset prior to the experiment. This will ensure that any electrical measurements obtained will be solely tissue derived.
Following mounting of the mucosa the tissue will be allowed to equilibrate before measurements of the tissues potential difference are made. If the tissue potential difference is satisfactory then the tissue will be placed under voltage clamp and the transepithelial resistance measured.
Tissue will be allowed to equilibrate under voltage clamp conditions before any experimental work is started.
Mucosal samples which pass the qualification pass/fail criteria will then progress to the study protocol.
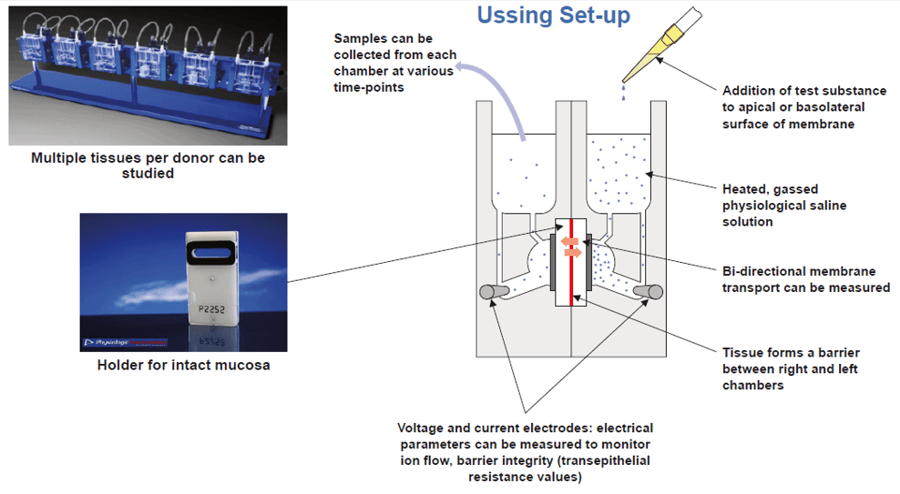
Ussing Chamber Methodology
To assess the metabolism of each test article, a single concentration of test article will be added to the apical chamber and samples taken from the apical and basolateral chambers at various time points between 0 and 120 minutes. Reference control compounds can also be run to allow direct comparison with test articles. An example of the conditions assessed for 3 test articles are detailed below:
- Test article 1
- Test article 2
- Test article 3
- Reference control compound(s)
Analysis
End point analysis can either be carried out in-house, outsourced or bath samples can be sent back to the sponsor.
Data from gastrointestinal metabolism assay
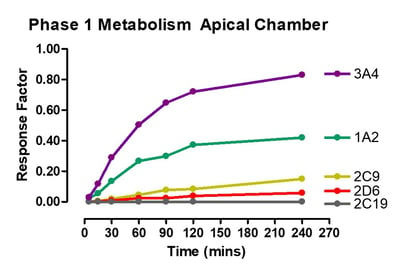
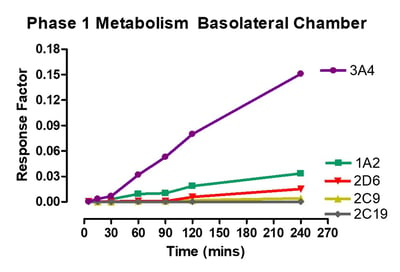
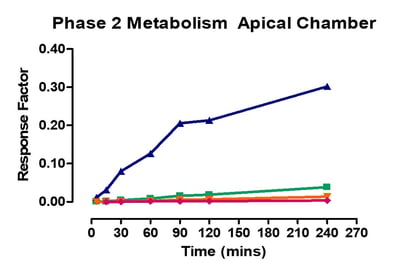
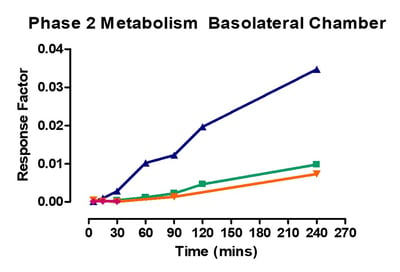
Phase 1 metabolism was measured by appearance of OH- Midazolam, OH- Tacrine, OH- Bufuralol, OH- Diclofenac, OH- Mephenytoin. Phase 2 metabolism was measured by appearance of 7-OH Coumarin Sulphate, 7-OH Coumarin Glucuronide, Diclofenac glucuronide and Hydroxy diclofenac glucuronide.


.jpg?width=756&height=425&name=Untitled%20design%20(10).jpg)