Laminins are a series of extracellular matrix (ECM) proteins found in all animals. These glycoproteins are a major component of the basal lamina layer of the basement membrane, which is a protein network that forms the foundation on which most cells and organ grow in the body. In vivo, laminins are important for regulating many biological processes, including adhesion, migration, and differentiation.1,2
Laminins are heterotrimeric proteins, consisting of an α chain, a β chain, and a γ chain in a 1:1:1 ratio.3 A total of 5 α chain isoforms, 3 β chain isoforms, and 3 γ chain isoforms are known, and the specific combination regulates the function of the particular laminin molecule. The different laminins are designated by the combination of isoforms present (Figure 1). For example, laminin-511 designates laminin composed of α5, β1, and γ1 subunits.
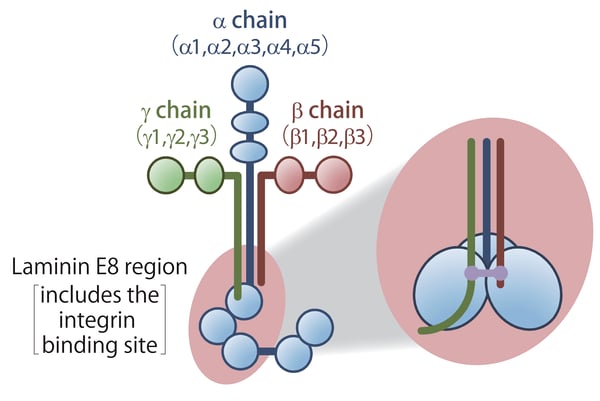
Figure 1. The structure of laminins. (Figure courtesy of Matrixome)
Laminins directly interact with integrin proteins in the cell membrane of animal cells. This interaction is one of the major components of the binding of cells to the basement membrane. Proteolytic studies of laminins have identified a protease-resistant core of the laminin trimer, called the E8 fragments, which contains the core integrin-binding domain (Figure 1).4 E8 fragments are ideal for in vitro experimentation because they are suitable for high-yield recombinant protein production, unlike the full-length laminins, which are challenging to express in vitro and difficult to form into active heterotrimers due to their large size and complex post-translational modification.
Recombinant human E8 fragments have been commercialized by several companies, including Matrixome (Osaka, Japan) because they are an ideal substrate for culturing a wide variety of cell types. Matrixome was founded based on studies showing that recombinant human laminin-511 E8 fragments, marketed under the name iMatrix-511 and available from REPROCELL, form an ideal matrix for the culture of human stem cells.5 Since the founding of Matrixome, a number of other isoforms of laminin E8 fragments have been introduced by Matrixome with a variety of cell specificities (Table 1).
| Integrin | ||
| iMatrix Product | Subunits | Cell Expression |
| iMatrix-111 | α6β1 α7X2β1 |
Liver Cells |
| iMatrix-211 | α7X2β1 | Cardiomyocytes Skeletal Muscle Cells |
| iMatrix-332 | α3β1 α6β4 |
Skin Corneal Epithelium |
| iMatrix-411 | α3β1 α6β1 |
Vascular Endothelium |
| iMatrix-511 iMatrix-511 SILK |
α3β1 α6β1 α6β4 |
Pluripotent Stem Cells Nerve Cells Retinal Pigment Cells Corneal Epithelium |
Table 1. Cell-specific integrin specificities for iMatrix laminins
iMatrix-511 Recombinant Human Laminin-511 E8 Fragments have been shown to bind tightly to Integrin α6β1, making it an ideal substrate for culturing human stem cells (Figure 2a and b).5 Original iMatrix-511 is expressed in CHO-S cells, while a new version, called iMatrix-511 SILK, is expressed in silkworm cocoons. iMatrix-511 SILK offers the same performance as the original iMatrix-511 at a lower price, and without risk of animal proteins.
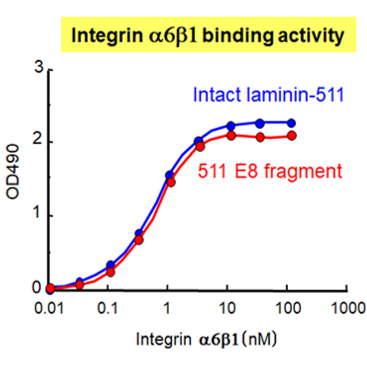 Figure 2a. Binding of integrin α6β1 protein to iMatrix-511. (Figure courtesy of Matrixome)
Figure 2a. Binding of integrin α6β1 protein to iMatrix-511. (Figure courtesy of Matrixome)
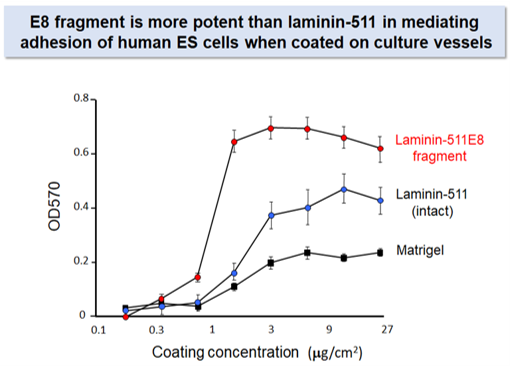
Figure 2b Binding of human ES cells to iMatrix-511. (Figure courtesy of Matrixome)
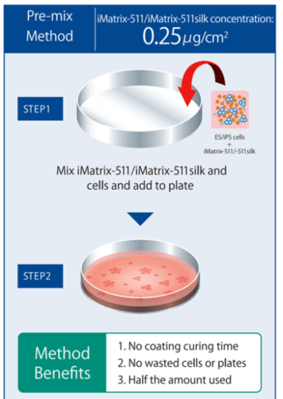
Figure 3. For convenience, iMatrix-511 from either source can be used with the Pre-mix Method by combining a cell suspension directly with iMatrix-511, without the need for pre-coating pates. (Figure courtesy of Matrixome)
Other iMatrix Laminin Products
- iMatrix 411: Laminin-411 is abundant in blood vessel basement membranes, and iMatrix-411 has been used to differentiate induced Pluripotent Stem Cells (iPSCs) into vascular endothelial cells and bile duct cells.6
- iMatrix-221: Laminin-211 is found in the basement membrane of muscle tissues. iMatrix-211 has been shown to be an excellent substrate for cardiomyocytes and skeletal muscle cells.
- iMatrix-332: Laminin-332 is present in keratinocytes and corneal cells. iMatrix-332 has recently been used as a key component for the differentiation and purification of corneal cells.7
- iMatrix-111: Laminin-111 is found in the liver. iMatrix-111 is an important component for the in vitro differentiation of iPSCs into hepatocytes.8
To find out more about iMatrix laminins, please visit the Cell Substrates section of our product cattalog.
References
-
- Timpl R, Rohde H, Robey PG, Rennard SI, Foidart JM, Martin GR “Lamini—Aglycoprotein from basement membrane.” J Biol Chem 254:9933 (1979).
- Durbeej M “Laminins.” Cell Tissue Res 339:259 (2010).
- Colognato H; Yurchenco PD “Form and function: The laminin family of heterotrimers.” Developmental Dynamics 218: 213 (2000)
- Goodman SL “α6β1 Integrin and laminin E8: An increasingly complex simple story.” Kidney International 41: 650 (1992).
- Miyazaki T; Futaki S; Suemori H; Taniguchi T; Yamada M; Kawasaki M; Hayashi M; Kumagai H; Nakasuji N; Sekiguchi K; Kawase E. “Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells.” Nature Commun 3: 1236 (2012)
- Ohta R; Niwa A; Taniguchi Y; Suzuki NM; Toga J; Yagi E; Saiki N; Nishinaka-Arai Y; Okado C; Watanabe A; Nakahata T; Sekiguchi K; Saito MK “Laminin-guided highly efficient endothelial commitment from human pluripotent stem cells.” Scientific Rep 6:35680 (2016).
- Shibata S; Hayashi R; Kudo Y; Okubo T; Imaizumi T; Katayama T; Ishikawa Y; Kobayashi Y; Toga K; Taniguchi Y; Honma Y; Sekiguchi K; Nichida K “Cell-type-specific adhesiveness and proliferation propensity on laminin isoforms enable purification of iPSC-derived corneal epithelium.” Stem Cell Rep 14: 663 (2020).
- Takayama K; Akita N; Mimura N; Akahira R; Taniguchi Y; Ikeda M; Sakurai F; Ohara O; Morio T; Sekiguchi K; Mizuguchi H “Generation of safe and therapeutically effective human induced pluripotent stem cell-derived hepatocyte-like cells for regenerative medicine.” Hepatol Commun 1: 1058 (2017).




%20(1).png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs)%20(1).png)
.jpg?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).jpg)
.png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).png)