Skin disease is one of the leading causes of global disease burden, affecting millions of people worldwide. Around 60% of people in the UK suffer from or have suffered from a skin disease at some point during their lifetime. Aging, environmental and genetic factors, and trauma can result in the development of a diverse range of skin diseases. Common examples include Psoriasis, Atopic Dermatitis (AD), Infection, and Skin Cancers. To stem the growth of skin disease globally, determining the cause of disease and developing effective treatments is key.
REPROCELL has developed a range of models using human skin to help researchers achieve this goal, summarized in the following article. If you have an interest in skin research or in developing novel compounds for the treatment of skin disease, download our skin brochure today to find out more.
What is skin tissue?
In humans, the largest organ in the body is the skin layer, accounting for up to 15% of the total body weight and a surface area of up to 2m2. It is composed of several layers — the epidermis, dermis, and the sub-cutaneous layer — each crucial to the many functions of the skin ranging from a protective barrier for internal tissues and organs, through regulation of body temperature, nutrient absorption, and the generation of vitamin D.
Full-thickness Skin Models
REPROCELL provides several assays using skin tissue that can facilitate both investigative research and the drug development process. We can access fresh healthy skin sections from our ethically-sourced Human Tissue Network. These tissues provide a rich source of subcutaneous resistance vessels for use in vascular studies or full-thickness biopsies for use in compound testing systems. We can also source biopsies from patients with skin conditions, including Psoriasis and AD.

Figure 1: Full-thickness punch biopsies can be made from healthy human skin to test the safety and efficacy of new compounds.
Skin models using healthy punch biopsies
Post-surgical tissues can be obtained for our healthy skin culture and induced phenotype models. These models are suited to researchers who want to measure the inflammatory or anti-inflammatory properties of more than one test article.
Healthy skin culture
Our healthy skin culture models use punch biopsies from residual surgical tissue to explore the inflammatory effects of a compound. By measuring inflammatory cytokine levels before and after drug addition, we can determine whether your article induces inflammation or not. Genetic expression of specific markers can also be measured using this assay.
Learn more about our healthy skin culture assay →
Inflamed skin model (PHA)
Our inflamed skin model assesses the anti-inflammatory properties of test compounds in vitro. Up to 20 different conditions per donor can be evaluated using biopsies prepared from fresh skin tissue. These healthy punch biopsies are induced to produce an inflammatory phenotype using phytohemagglutinin (PHA).
Learn more about our inflamed skin model →
Skin culture induced psoriasis model
Our induced psoriasis model replicates the activated Th17 pathway associated with this disease. The model allows topical or systemic administration of compounds to skin biopsies prepared from fresh tissue and the subsequent measurement of inflammatory responses.
Learn more about our induced psoriasis model →
Induced atopic dermatitis model
Similar to the other induced models, our induced AD model uses healthy skin punch biopsies induced to display an AD phenotype. Using a propriety cocktail, we activate the TH2 pathway to reflect the pathology of AD, and then measure the anti-inflammatory effects of any compounds added.
Learn more about our induced atopic dermatitis model →
Skin culture induced acne model (LPS)
For our induced acne model, lipopolysaccharide (LPS) is used to activate the TLR2/TLR4 pathway in healthy punch biopsies. Measuring cytokine release can then be used to estimate the anti-inflammatory effects of your test articles.
Learn more about our induced acne model →
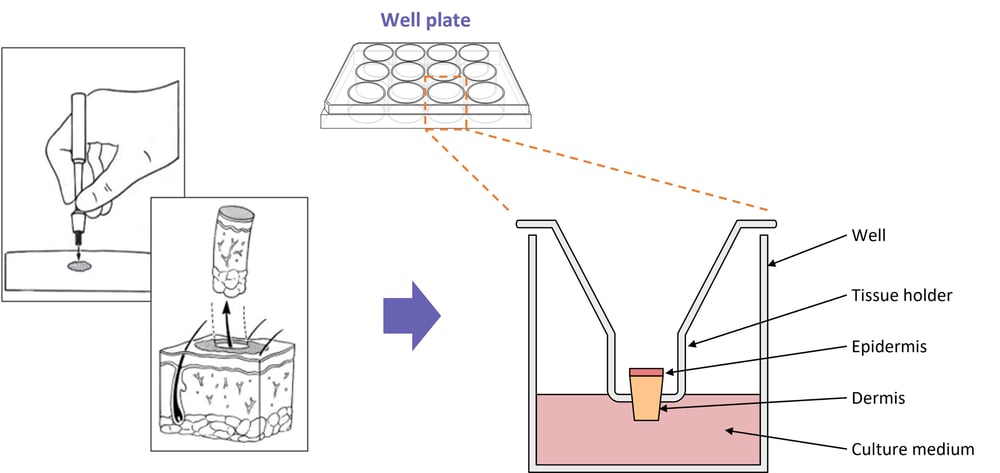
Figure 2: Diagram showing a typical setup for our skin models using healthy punch biopsies.
Skin culture models using diseased patient biopsies
Clinical biopsies from patients living with skin disease can also be procured via our clinical network. These models are best suited to researchers looking to assess the efficacy of one or two lead compounds. Up to three biopsies can be obtained per donor with psoriasis or AD.
Skin Biopsies from Atopic Dermatitis Donors
This model uses fresh skin tissue from patients with AD. It is the closest model to measuring compound efficacy in vivo, so is typically used for lead compounds that are almost ready for clinical trial. Our researchers assess whether the compound causes a reduction in inflammatory cytokine release with Betamethasone as a reference.
Learn more about our skin biopsies from AD donors →
Skin Biopsies from Psoriasis Donors
Skin biopsies from donors with psoriasis can be used to test the anti-inflammatory effects of your compounds. As with our AD model, this assay is typically used for optimizing lead compounds just before clinical trial and uses Betamethasone as a reference compound.
Learn more about our skin biopsies from psoriasis donors →
Bioengineered 3D Human Skin In Vitro Models
If you are looking for a translational but more high-throughput model, our bioengineered 3D human skin in vitro models may be the perfect solution. Full-thickness punch skin biopsies from both healthy and diseased donors can be used for the establishment of this model. Keratinocyte and fibroblast primary cell cultures are isolated from these biopsies and then co-cultured on Alvetex scaffolds at the air-liquid interface. You can find out more about the establishment of this model here.
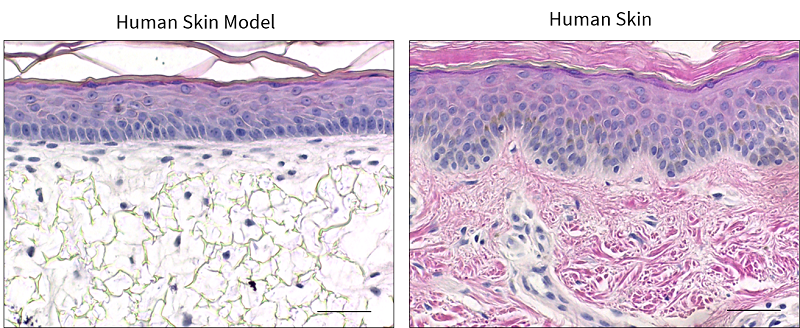
Figure 3: Histochemical analysis of our in vitro 3D human skin model compared with native human skin.
Vascular Assays
Our vascular assays assess the effect of compounds on the functional capability of blood vessels, including vasoconstriction, vasodilation, vascular resistance, and vascular permeability. Our staff expertly dissect arteries and veins from tissue for analysis in our organ bath and myograph systems. Subcutaneous resistance arteries from fresh skin tissue are a valuable resource for these assays — you can find out more about our cardiovascular assays here.
Editor's note: This blog was first published in November 2018 and has since been updated for accuracy and clarity



%20(1).png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs)%20(1).png)
.jpg?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).jpg)
.png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).png)