Example Protocol for the Culture of the Caco-2 Cell Line on Alvetex® Scaffold in Well Insert and Well Plate Formats With or Without a Collagen I Gel Layer
● Download this protocol as a PDF (3.9 MB)
1. Introduction
Caco-2 is a human colorectal adenocarcinoma cell line isolated in the late 1970s from colon carcinoma tissue[1]. Although Caco-2 cells have been used to model colon cancer cytotoxicity[2], they are better known for their ability to differentiate into functional enterocytes in vitro, characterised by the presence of tight junctions, a brush border and brush-border-associated hydrolases[3]. This has led to their widespread use as in vitro models of intestinal drug absorption[4] and bacterial or viral gut infection[5,6].
Alvetex Scaffold is available in several cell culture formats including 24 well plate (AVP006), 12 well plate (AVP002), 6 well insert (AVP004), 12 well insert (AVP005), and 24 well insert (AVP012).
24 well and 12 well plates are suitable for shorter term cultures and for applications where limited cell penetration into the scaffold is required. Well insert formats generally support longer term cultures and deeper cell penetration into the scaffold. They also provide for conveniently tailored media set ups (see the Alvetex Scaffold Quick Start protocol).
The availability of two different well insert formats enables choice on the basis of desired culture size and cell expenditure. 6 well inserts can be placed in conventional 6 well plates, while 12 well inserts can be placed in either 6 well plates or 12 well plates, depending on media requirements. Alternatively, both insert types can be housed in the dedicated Well Insert Holder in Deep Well Petri Dish (AVP015) to allow for increased media volumes and prolonged cell culture.
2. Methods
2.1. Preparation for 3D Cell Culture on Alvetex Scaffold
- Caco-2 cells (ATCC, HTB-37) were routinely maintained in T-75 flasks.
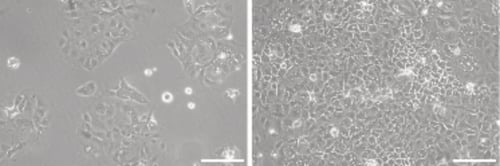
Figure 1. Phase contrast micrograph of Caco-2 cells grown in conventional 2D culture plates. Image shows cells at low (left) and high (right) confluency. Scale bars: 100 µm.
-
Complete growth media consisted of: DMEM High glucose supplemented with 20 % v/v FBS, 0.1mM nonessential amino acids, 10 mM HEPES buffer, 2 μm L-glutamine and 100 U/mL Penicillin/Streptomycin.
-
Cells were harvested by trypsinisation and centrifuged for 5 minutes (1000 rpm). The supernatant was discarded and the cell pellet was re-suspended in an appropriate volume of media for cell counting by Trypan Blue.
-
Cells were re-suspended at a concentration of 1.0 × 107 cells/mL for seeding.
2.2. Collagen I Gel Top Layer on Alvetex Scaffold
A detailed protocol describing how to coat the top surface of Alvetex Scaffold with Collagen I can be found at Alvetex Protocols.
2.3. 12 well Plate Format (AVP002)
-
Alvetex Scaffold 12 well plates were prepared for seeding with a 70 % ethanol wash (4 mL per well) and subsequent media washes (twice with 4 mL of media each).
-
50 µL of the cell suspension was added to the centre of the Alvetex Scaffold disc, which was equivalent to 0.5 × 106 cells per well.
-
The plate was incubated for at least 1 hour at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
-
4 mL of media was added to each well taking care not to dislodge cells from the Alvetex Scaffold.
-
Plates were re-incubated and maintained by complete media exchange every other day.
Note: This method can be applied to the use of Alvetex Scaffold in 24 well insert format (AVP006). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start Protocol.
2.4. 6 well Insert Format (AVP004)
-
Alvetex Scaffold 6 well inserts in 6 well plate format were prepared for seeding by dipping in 70 % ethanol followed by media washes (twice with 10 mL per well).
-
Wells were filled from the outside of the insert with enough medium to allow it to rise inside the insert and cover the substrate, but not to go over the sides of the inserts, i.e. 7 ± 1 mL, as described for feeding above and below separately in the Alvetex Scaffold Quick Start Protocol.
-
50 μL of the cell suspension was added in droplets over the surface of the Alvetex Scaffold disc, which was equivalent to 0.5 × 106 cells per well.
-
The plate was incubated overnight at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
-
Media was added to each well to a total volume of 10 mL the following morning taking care not to dislodge cells from the Alvetex Scaffold.
-
Plates were re-incubated and maintained by complete media exchange every other day.
Note: This method can be applied to the use of Alvetex Scaffold in 12 well insert format (AVP005). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start Protocol.
3. Example Data
3.1. 12 well plate format, AVP002
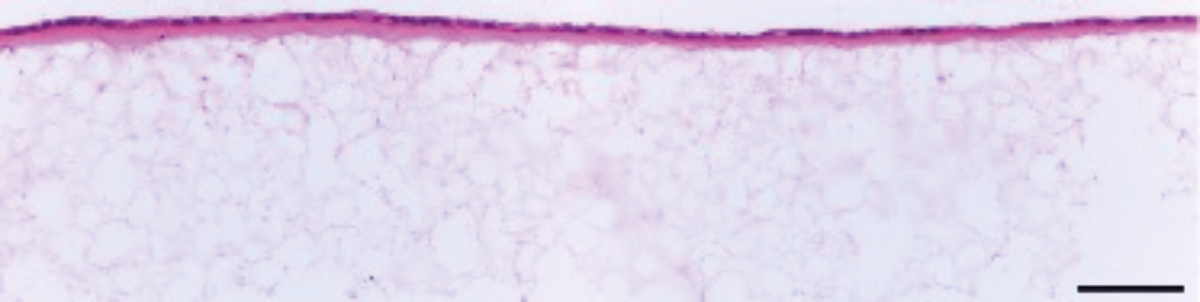
Figure 2. Brightfield micrograph showing the structure of Caco-2 cells cultured with a Collagen I gel layer for 7 days on 22 mm diameter Alvetex Scaffold discs presented in 12-well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 μm) and counterstained with haematoxylin and eosin. Scale bar: 100 µm.
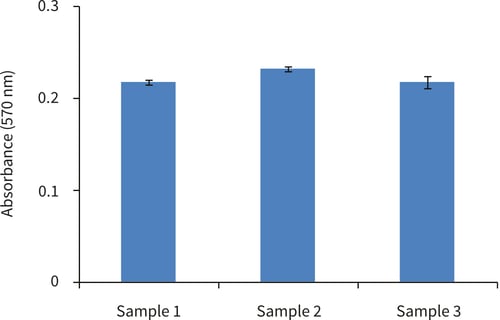
Figure 3. Biochemical analysis of cell viability using a standard MTT assay. Data from 3 sample replicates of Caco-2 cells are shown, sampled in triplicates (n=3, mean ± SD). Cells were cultured with a Collagen I gel top layer for 3 days on 22 mm Alvetex Scaffold discs presented in 12 well plate format.
3.2. 6 well insert format, AVP004

Figure 4. Brightfield micrograph showing the structure of Caco-2 cells cultured with a Collagen I gel top layer for 7 days on 22 mm diameter Alvetex Scaffold discs presented in 6-well insert in 6 well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 μm) and counterstained with haematoxylin and eosin.
Scale bar: 100 µm.
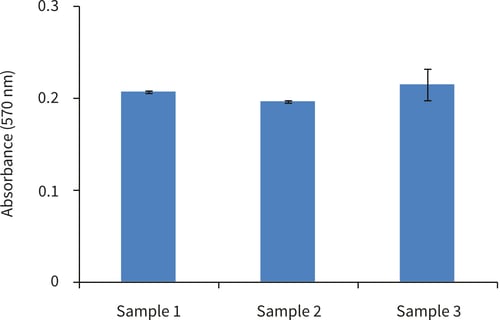
Figure 5. Biochemical analysis of cell viability using a standard MTT assay. Data from 3 sample replicates of Caco-2 cells are shown, sampled in triplicates (n=3, mean ± SD). Cells were cultured with a Collagen I gel top layer for 3 days on 22 mm Alvetex Scaffold discs presented in 6-well inserts in 6 well plate format.
3.3. 6 well insert format, AVP004, with or without a collagen I layer
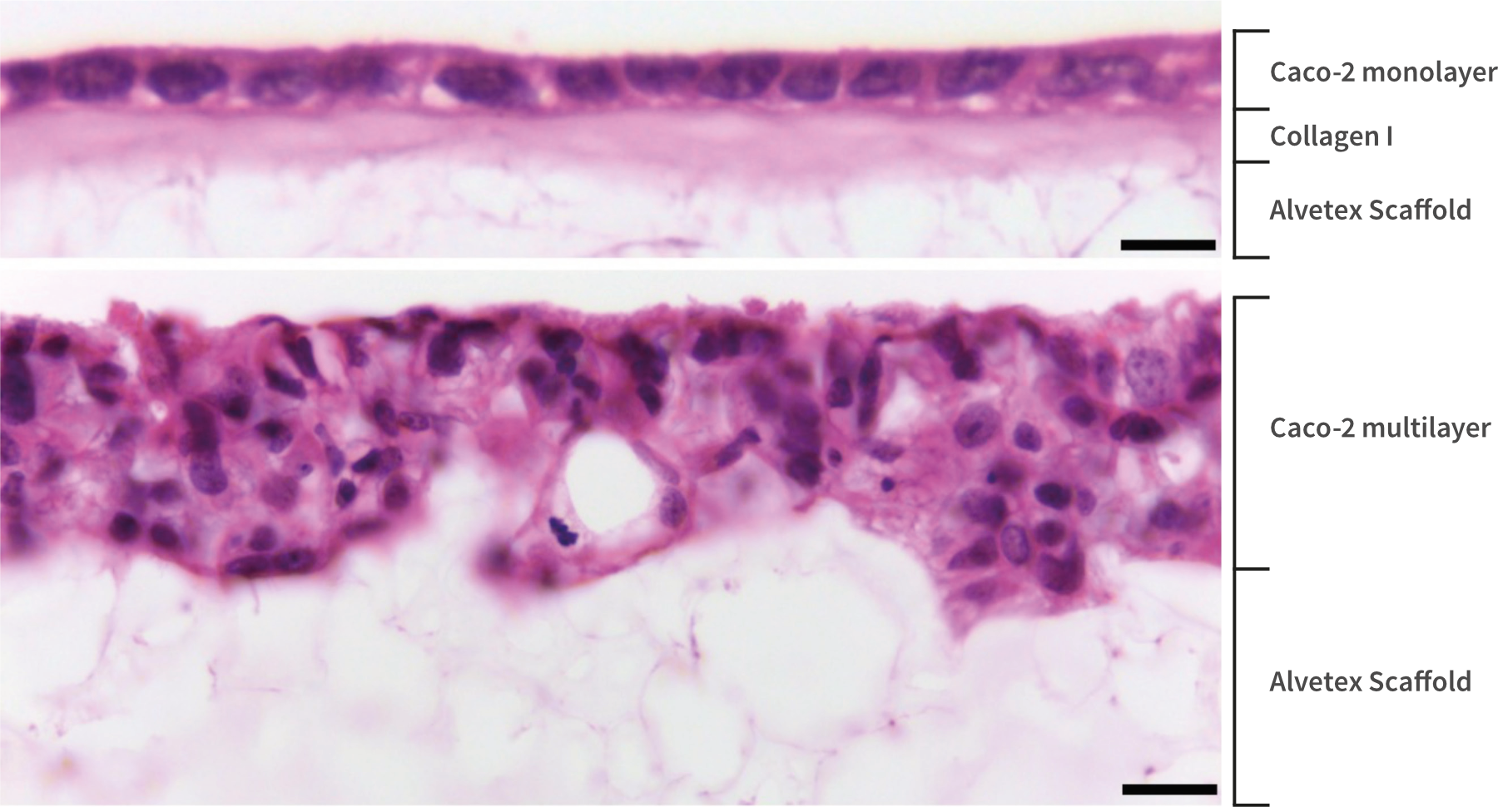
Figure 6. High magnification brightfield micrographs showing the structure of Caco-2 cells cultured with (top) or without (bottom) a Collagen I gel top layer for 7 days on 22 mm diameter Alvetex Scaffold discs presented in 6 well insert in 6 well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 µm) and counterstained with haematoxylin and eosin. Scale bars: 10 µm (top) and 25 µm (bottom).
4. References
-
Fogh J et al, 1977. One hundred and twenty-seven cultured human tumor cell lines producing tumors in nude mice. J Natl Cancer Inst 59(1): 221-6.
-
Viraq P et al, 2012. Superior cytotoxicity and DNA cross-link induction by oxaliplatin versus cisplatin at lower cellular uptake in colorectal cancer cell lines. Anticancer Drugs 23(10): 1032- 8.
-
Chantret I et al, 1988. Epithelial polarity, villin expression, and enterocytic differentiation of cultured human colon carcinoma cells: a survey of twenty cell lines. Cancer Res 48(7): 1936- 42.
-
Balimane PV et al, 2006. Current industrial practices of assessing permeability and P- glycoprotein interaction. AAPS J 8(1): E1-13.
-
Gaillard JL et al, 1987. In vitro model of penetration and intracellular growth of Listeria monocytogenes in the human enterocyte-like cell line Caco-2. Infect Immun 55(11): 2822-9.
-
Svensson L et al, 1991. Symmetric infection of rotavirus on polarized human intestinal epithelial (Caco-2) cells. J Virol 65(8): 4190-7.