Relaxation in Human Uterus (Adrenoceptor – Isoprenaline)
Drug Discovery Assay – reference number: B082
Overview
| Assay type: | GU |
| Tissue: | Human uterus (healthy) |
| Target: | Adrenoceptor |
| Control compound: | Isoprenaline |
| Study type: | Organ bath |
| Functional endpoint: | Relaxation |
Assay Description
This assay assesses whether test articles cause a reduction in spontaneous activity in isolated human uterine tissue, with isoprenaline as a reference compound.
The uterus is one of the female reproductive organs. The main functions of the uterus are to provide a suitable site for an implanting blastocyst, to expand with the growing foetus and to contract strongly to expel the foetus. Alterations to the anatomy of the uterus may adversely affect its functionality.
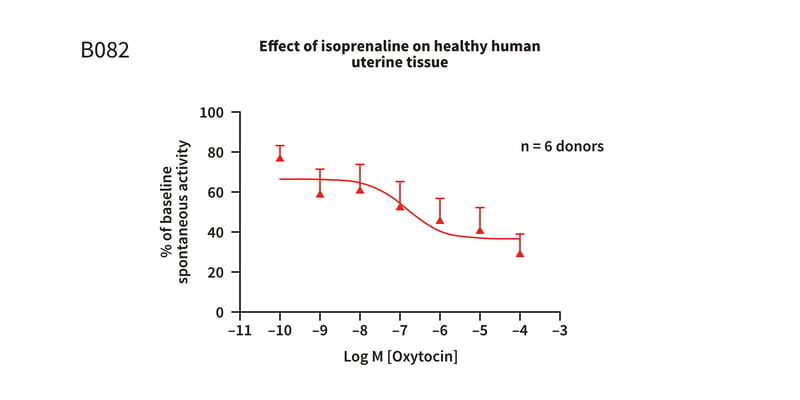 Figure 1: Cumulative concentration response curve to isoprenaline in healthy human uterine tissue, pre-constricted with EC80oxytocin, demonstrating a concentration dependent reduction in the spontaneous activity of the tissue. The Log EC50 = −6.9 ± 0.72M (141.9nM) (mean ± S.E.M), with a maximum reduction of 71.14 ± 9.69%.
Figure 1: Cumulative concentration response curve to isoprenaline in healthy human uterine tissue, pre-constricted with EC80oxytocin, demonstrating a concentration dependent reduction in the spontaneous activity of the tissue. The Log EC50 = −6.9 ± 0.72M (141.9nM) (mean ± S.E.M), with a maximum reduction of 71.14 ± 9.69%.
Testing Information
Introduction
The specific results that will be provided are the effects of increasing concentrations of test articles on the spontaneous activity of isolated human uterine muscle.
Test Article Requirements
Test article(s) to be provided by the Sponsor in storable aliquots at required test concentrations with information on diluent vehicle used. Stock solutions are prepared in distilled water unless otherwise requested. Bath volumes are 25mL; sponsor to provide sufficient test article to run the entire study.
Suggested Testing
In duplicate at 6 concentrations.
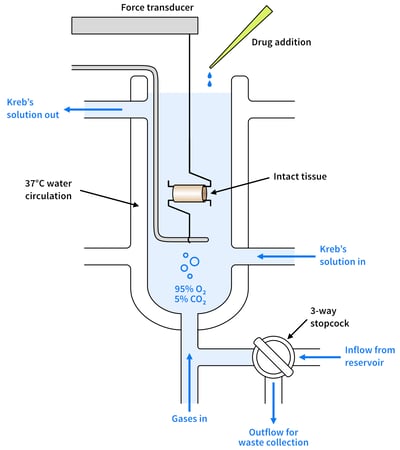 Figure 2: A diagram showing organ bath set-up
Figure 2: A diagram showing organ bath set-up
Study Outline
Rationale and Experimental Design
This assay assesses whether test articles cause a reduction in spontaneous activity in isolated human uterine tissue, with isoprenaline as a reference compound.
Exclusion Criteria
No specific exclusion criteria are in place other than to reject macroscopically diseased/necrotic tissue. Furthermore, tissues which do not respond to the standard pharmacology checks will be excluded.
Standardisation and Qualification
All individual muscle strips are initially processed through standardisation and qualification procedures to ensure functionality, prior to starting the study protocol.
Muscle strips are processed through a standardisation procedure to reduce signal variability prior to pharmacological intervention. This ensures that muscle strips are maintained under appropriate physiological tension throughout the experiments.
Muscle strips which show stable regular spontaneous contractions will the progress to the study protocol.
Smooth Muscle Contractility Assay
Agonist Relaxation Assays
To assess the ability of each article to reduce spontaneous activity of the tissue, 6 point cumulative concentration response curves will be performed for each test article. A positive control compound and representative test article vehicle CCRC will also be run to allow direct comparison with test articles.
An example of the conditions assessed for 3 test articles are detailed below (each condition will be run in duplicate muscle strips):
-
Representative test article vehicle CCRC
-
Test article 1 CCRC
-
Test article 2 CCRC
-
Test article 3 CCRC
-
Positive control CCRC
Supplementary Option
To assess the involvement of a specific receptor subtype in any observed responses, the concentration of test article eliciting the largest response can subsequently (following a wash out and recovery period) be tested in the presence of a specific antagonist. This supplementary option will incur an extra charge.
Antagonist Relaxation Assays
Option 1 − IC50 Determination
To assess the ability of each test article to antagonise an agonist mediated reduction in spontaneous activity, 6 point cumulative concentration response curves will be performed for each test article. These concentration response curves (CCRC’s) will be performed following incubation with the appropriate reference agonist. A positive control compound and representative test article vehicle CCRC will also be run to allow direct comparison with test articles.
An example of the conditions assessed for 3 test articles are detailed below (each condition will be run in duplicate muscle strips):
-
Representative test article vehicle CCRC
-
Test article 1 CCRC
-
Test article 2 CCRC
-
Test article 3 CCRC
-
Positive control CCRC
Option 2 − pA2 Determination
To assess the ability of a test article to antagonise an agonist mediated reduction in spontaneous activity; 6 point cumulative concentration response curves (CCRC’s) will firstly be performed for the reference agonist. Following a wash out and recovery period, muscle strips will be incubated with the test article (3 different concentrations of test article will be assessed), positive control or test article vehicle before the same 6 point cumulative concentration response curve will be repeated for the reference agonist.
An example of the conditions assessed for 3 test articles are detailed below (each condition will be run in duplicate muscle strips):
-
Representative test article vehicle
-
Test article 1 concentration 1
-
Test article 1 concentration 2
-
Test article 1 concentration 3
-
Test article 2 concentration 1
-
Test article 2 concentration 2
-
Test article 2 concentration 3
-
Test article 3 concentration 1
-
Test article 3 concentration 2
-
Test article 3 concentration 3
-
Positive control
Analysis
Responses shall be expressed as tension over a specified period of time (g sec) or as a % reduction in baseline spontaneous activity. Statistical analysis will be performed (where appropriate) using GraphPad Prism, with the results being shown in graphical form in the final report.
.jpg?width=756&height=425&name=New%20Approach%20Methodologies%20(NAMs).jpg)

