Modulation of Neuronally Induced Contractions (EFS) of Human Bladder (Buscarinic and β-Adrenergic Receptors – Carbachol, Isoprenaline)
Drug Discovery Assay – reference number: B108
Overview
| Assay Type: | GU |
| Tissue: | Human bladder (healthy) |
| Target: | Muscarinic/β-adrenergic receptors/ion channels |
| Control compound: | Carbachol/isoproterenol/retigabine |
| Study type: | Organ bath |
| Functional endpoint: | Modulation of EFS-induced contractions |
Assay Description
This assay assesses whether test articles can modify EFS-induced contractions in human isolated bladder strips, with carbachol/isoproterenol/retigabine as reference compounds.
The primary function of the bladder is the storage of urine before micturition. The contraction and relaxation of the smooth muscle controls the emptying and filling of the bladder. Electrical field stimulation selectively activates prejunctional nerves to release neurotransmitters leading to contraction. Alterations to bladder contractility can exacerbate or improve bladder dysfunction including overactive/incontinence, nocturia, residual urine, etc.
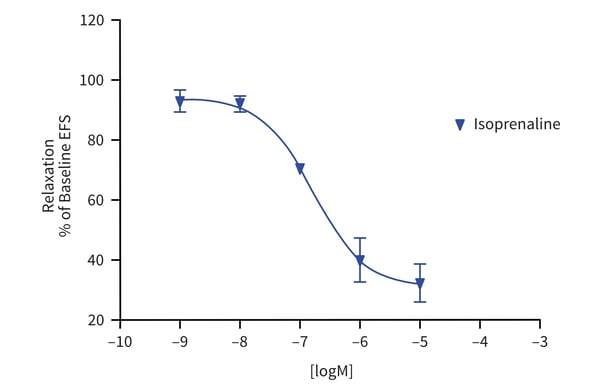
Figure 1: Cumulative concentration response curve to isoproterenol (or vehicle) on EFS-induced contractions in human isolated bladder.
Testing Information
Introduction
The specific results that will be provided are the effects of increasing concentrations of test articles on EFS-induced contractile responses of human isolated bladder muscle.
Test Article Requirements
Test article(s) to be provided by the Sponsor in storable aliquots at required test concentrations with information on diluent vehicle used. Stock solutions are prepared in distilled water unless otherwise requested. Bath volumes are 25mL; sponsor to provide sufficient test article to run the entire study.
Suggested Testing
In duplicate at 6 concentrations.
Study Outline
Rationale and Experimental Design
This assay assesses whether test articles can modify neuronally-mediated contractions in isolated human bladder strips, with carbachol/isoproterenol as reference compounds.
Exclusion Criteria
No specific exclusion criteria are in place other than to reject macroscopically diseased/necrotic tissue. Furthermore, tissues which do not respond to the standard pharmacology checks will be excluded.
Standardisation and Qualification
All individual muscle strips are initially processed through standardisation and qualification procedures to ensure functionality, prior to starting the study protocol.
Muscle strips are processed through a standardisation procedure to reduce signal variability prior to pharmacological intervention. This ensures that muscle strips are maintained under appropriate physiological tension throughout the experiments.
Following standardisation, the muscle strips are stimulated with EFS to ensure robust contractile responses.
Muscle strips that pass the standardisation and qualification pass/fail criteria will then progress to the study protocol.
Contractility Assay
Modulation of EFS-induced contractions
To assess the ability of each test article to modulate EFS-induced contractility, 6 point cumulative concentration response curves (CCRCs) will be performed for each test article. A positive control compound (and vehicle) CCRC will also be run to allow direct comparison with test articles.
An example of the conditions assessed for 3 test articles are detailed below (each condition can be run in duplicate muscle strips):
- Representative test article vehicle CCRC
- Test article 1 CCRC
- Test article 2 CCRC
- Test article 3 CCRC
- Positive control CCRC
Analysis
Responses shall be expressed either in milligrams or grams tension, or as a % of baseline or maximum response. Statistical analysis will be performed (where appropriate) using GraphPad Prism, with the results being shown in graphical form in the final report.
.jpg?width=756&height=425&name=New%20Approach%20Methodologies%20(NAMs).jpg)

