Most biologists are familiar with the precision gene editing system CRISPR-Cas9. But what about CRISPR-SNIPER†? This advanced gene editing system uses droplet digital PCR (ddPCR)[1] to enhance your iPSC research, saving you time, money and effort.
What is CRISPR-SNIPER?
CRIPSR-SNIPER is an advanced CRISPR-Cas9 gene editing system that uses ddPCR[1] and culture-based screening technology, enabling multiplex gene modification, biallelic gene insertions and single nucleotide variations (SNV). It uses a checkerboard of culture conditions to determine the most effective gRNA transfection conditions prior to transfection and cloning. The technique was pioneered by Dr Tsukasa Sugo, the founder of GenAhead Bio[2], and carries freedom to operate for research use, including iPSC gene editing.
Inserting large genes
If you are a researcher working in developmental biology, you may want to track stem cell differentiation using fluorescent tags. However, these genes are often difficult to insert using conventional CRISPR gene editing, as knock-in (KI) efficiency declines when an insert exceeds 2 kbp.
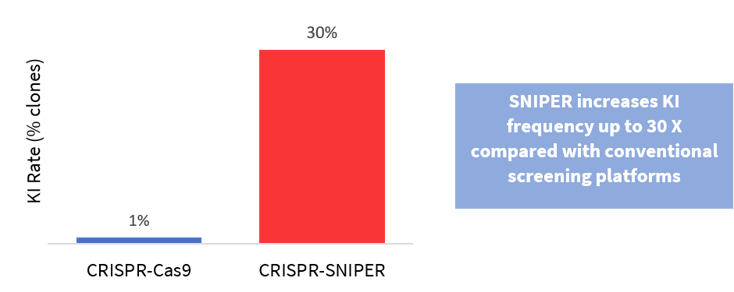
With CRISPR-SNIPER's increased KI rate, it is easier to insert large functional gene segments up to 7 kbp in size. This is because CRISPR-SNIPER uses advanced screening methods which can achieve up to 30 times* the detection rate compared with conventional gene editing techniques.
Creating your desired genotype
If the phenotype of your desired iPSC mutation varies dependent on whether the gene is heterozygous or homozygous, you may struggle to achieve your desired edit using CRISPR-Cas9 alone. With CRISPR-SNIPER, both DNA strands can be modified accurately to obtain your desired genotype – enabling the creation of both homozygous and heterozygous iPSC cell lines with ease.
Increasing screening specificity
If you are looking to obtain iPSCs with a single point mutation (SNV) you will be happy to find out that these edits are also easier and with CRISPR-SNIPER. This is because CRISPR-SNIPER evaluates gene editing efficacy under multiple culture conditions and uses nickase to increase editing efficacy; meaning sequences that are similar to your gene of interest are excluded during the screening process.
Editing multiple genes simultaneously
If establishment of your iPSC model involves multiple modifications, you will need to perform several sequential gene editing experiments. This is not only laborious, but can increase the passage number of your iPSC lines significantly.
As CRISPR-SNIPER allows rapid optimization of gene editing conditions, this advanced gene editing technique makes it possible to modify multiple genes simultaneously. This is achieved by optimizing the culture conditions for a multiplex panel of gRNAs targeted towards your chosen genes, and then using this cocktail to edit your cells in a single experiment. By editing multiple genes at once, you can reduce the negative effects that accompany extended passaging and culture your iPS cells for longer.
CRISPR-SNIPER is led by the iPSC experts
REPROCELL is the only iPSC company able to offer CRISPR-SNIPER gene editing. Where most gene editing companies specialize in knock-out (KO) modifications, we have substantial experience in SNVs, KI modifications and iPSC editing. With over a decade of experience in iPSC editing and over 70 CRISPR-SNIPER projects completed to date, you can trust our scientists to deliver the iPSCs you need, when you need them.
Want to find out more?
Did we provide a solution to your gene editing needs today? If not, let us know. If you are interested in finding out more about CRISPR-SNIPER service, download our CRISPR-SNIPER guide today.
You can also explore our free online resources to learn more about the most advanced iPSC gene editing service on the market.
- Related post: How to determine gene editing success in 2 weeks [BLOG]
†SNIPER is the acronym for Specification of Newly Integrated Position and Exclusion of Random-Integration
*For SNP modification, SNIPER screening detects positive clones at a 30× higher frequency (30%) compared with conventional screening (1%)
Note: The Modified Cells are developed, manufactured or supplied by GenAhead Bio Inc. under license from ERS Genomics Limited.
Further reading
- Puckrin, Zara; Annand, Robert & McAulay, Karen. (2020). The solution to complex CRISPR Modifications: Digital droplet PCR. 30.
- GenAhead Bio website
Editors note: This post was originally published in June 2019 and has been since updated for accuracy and clarity.
.jpeg?width=1280&height=720&name=14NOV19%20Gene%20editing%20stock%20image%20(1).jpeg)