Example Protocol for the Culture of Primary Rat MSCs on Alvetex® Scaffold in Well Insert Formats
● Download this protocol as a PDF (0.9 MB)
1. Introduction
Alvetex Scaffold is available in several cell culture formats including 24 well plate (AVP006), 12 well plate (AVP002), 6 well insert (AVP004), 12 well insert (AVP005), and 24 well insert (AVP012).
24 well and 12 well plates are suitable for shorter term cultures and for applications where limited cell penetration into the scaffold is required. Well insert formats generally support longer term cultures and deeper cell penetration into the scaffold. They also provide for conveniently tailored media set ups (see the Alvetex Scaffold Quick Start protocol).
The availability of two different well insert formats enables choice on the basis of desired culture size and cell expenditure. 6 well inserts can be placed in conventional 6 well plates, while 12 well inserts can be placed in either 6 well plates or 12 well plates, depending on media requirements. Alternatively, both insert types can be housed in the dedicated Well Insert Holder in Deep Well Petri Dish (AVP015) to allow for increased media volumes and prolonged cell culture.
2. Methods
2.1. Preparation for 3D Cell Culture on Alvetex Scaffold
- Primary rat MSCs were derived from the bone marrow of 6-8 week old male Wistar rats (Harlan) using standard, previously published, methodology[1], and used at P1 for experiments. MSCs were routinely maintained in T-75 flasks.
- Complete growth media consisted of: DMEM High glucose supplemented with 10 % v/v heat inactivated FBS, 1× non-essential amino acids, 2 μm L-glutamine and 100 U/mL Penicillin/ Streptomycin.
- Cells were harvested by trypsinisation and centrifuged for 5 minutes (1000 rpm). The supernatant was discarded and the cell pellet was re-suspended in an appropriate volume of media for cell counting by Trypan Blue.
- 4. Cells were re-suspended at a concentration of 1.0 × 107 cells/mL for seeding.
2.2. 6-well Insert Format (AVP004)
- Alvetex Scaffold 6-well inserts in 6-well plate format were prepared for seeding by dipping in 70 % ethanol and washed twice with media (7 mL per well).
- 100 μL of the cell suspension was added to the centre of the Alvetec Scaffold disc, which was equivalent to 1 × 106 cells per well.
- The plate was incubated for at least 15 minutes at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
- 10 mL of media was added to each well taking care not to dislodge cells from Alvetex Scaffold.
- Plates were re-incubated and maintained by complete media exchange after every 2-3 days.
Note: This method can be applied to the use of Alvetex Scaffold in 12-well insert format (AVP005). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start protocol.
3. Example data
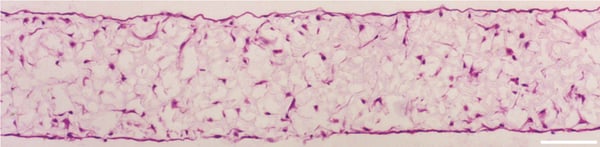
FIGURE 1. Brightfield micrographs showing the structure of primary rat MSCs cultured for 14 days on 22 mm diameter Alvetex Scaffold discs presented in 6-well insert in 6-well plate format. Cells were fixed, embedded in paraffin wax, sectioned (10 µm) and counterstained with haematoxylin and eosin. Scale bar 100 µm.
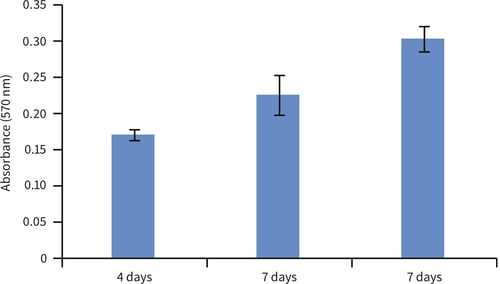
FIGURE 2. Biochemical analysis of cell viability using a standard MTT assay. For each timepoint, data from 3 sample replicates of primary rat MSCs are shown (n=3, mean ± SEM). Cells were cultured for 4, 7 and 14 days on 22 mm Alvetex Scaffold discs presented in 6-well insert in 6-well plate format.
4. Reference
- Croft, A.P. and S.A. Przyborski, Formation of neurons by non-neural adult stem cells: potential mechanism implicates an artifact of growth in culture. Stem Cells, 2006. 24(8): p. 1841-51.