Example Protocol for the Culture of the PC-3 Cell Line on Alvetex® Scaffold in Well Plate and Well Insert Formats
● Download this protocol as a PDF (3.2 MB)
1. Introduction
The PC-3 cell line is one of the main cell lines used in prostate cancer research. PC-3 cells have a high metastatic potential compared to other prostate cancer cell lines such as LNCaP and DU145. This cell line was established in 1979 from a bone metastasis site of a 62 year old Caucasian male.[1] PC-3 cells are reported to be androgen receptor (AR) negative. However there are contradicting reports suggesting that PC-3 cells express AR mRNA.[2] They are also known not to express Prostate Specific Antigen and are PSMA (Prostate-Specific Membrane Antigen) negative.
2. Methods
2.1. Preparation for 3D cell culture on Alvetex Scaffold
- PC-3 cells (ATCC, CRL-1 435) were routinely maintained in T-75 flasks.
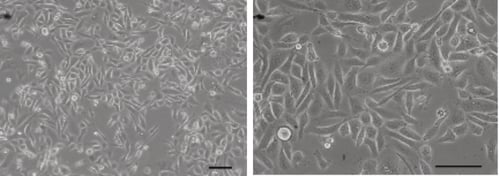
Figure 1. Phase contrast micrograph of PC-3 cells grown in conventional 2D culture plates. Scale bars: 100 µm.
-
Complete media consisted of: Either ATCC formulated F12-K medium (Kaighn’s modification) (Invitrogen (Thermo Fisher), 21127-022) supplemented with 10 % v/v FBS and 100 U/mL Penicillin/ Streptomycin or F12-K medium (Kaighn’s modification) supplemented with 10 % v/v FBS and 100 U/mL Penicillin/ Streptomycin.
-
Cells were harvested by trypsinisation and centrifuged for 5 minutes (1200 rpm). The supernatant was discarded and the cell pellet was re-suspended in an appropriate volume of media for cell counting by Trypan Blue.
-
Follow one of the below methods according to your choice of Alvetex Scaffold format.
2.2. 12 well plate format (AVP002)
-
Cells were re-suspended at a concentration of 2 × 106 cells/mL, 4 × 106 cells/mL and 8 × 106 cells/mL.
-
Alvetex Scaffold 12 well plates were prepared for seeding with 70 % ethanol (2 mL per well) and media washes (twice with 3 mL of media each) as described in the product information leaflet.
-
125 µL aliquots of the cell suspensions were added to the centre of the Alvetex Scaffold disc to give cell seeding numbers of 0.25 × 106, 0.5 × 106 and 1 × 106 cells per disc.
-
The plate was incubated for 60 min at 37 °C with 5 % CO2 to allow the cells to settle.
-
4 mL of culture medium was added to each well taking care not to dislodge the cells from Alvetex Scaffold.
-
Medium changes were performed every 2 days.
Note: This method can be applied to the use of Alvetex Scaffold in 24 well plate format (AVP006). Adjust cell seeding and media volumes according to the guidelines provided in the Alvetex Scaffold Quick Start protocol.
2.3. 6 well insert format (AVP004)
-
Cells were re-suspended at a concentration of 4 × 106 cells/mL, 8 × 106 cells/mL and 16 × 106 cells/mL.
-
Alvetex Scaffold 6 well inserts were prepared for cell culture by washing in 70 % ethanol, 1 × PBS and PC-3 complete culture medium.
-
125 µL aliquots of the cell suspensions were added to the centre of the Alvetex Scaffold disc to give cell numbers of 0.5 × 106, 1 × 106 and 2 × 106 cells per disc accordingly.
-
The plates were incubated for 60 min at 37 °C with 5 % CO2 to allow the cells to settle into the scaffold.
-
9 mL of culture medium was added to each well taking care not to dislodge the cells from Alvetex Scaffold.
-
Medium changes were performed every 2 days.
Note: This method can be applied to the use of Alvetex Scaffold in 12 well insert format (AVP005). Adjust cell seeding and media volumes according to the guidelines provided inthe Alvetex Scaffold Quick Start protocol.
96 well plate format (AVP009)
-
Cells were re-suspended at a concentration of 125,000 cells/mL, 250,000 cells/mL and 500,000 cells/mL for seeding the appropriate cell numbers in 200 μL total medium volume.
-
Alvetex Scaffold 96 well plates were prepared for cell culture by washing the wells with 100 μL of 70 % ethanol, 200 μL of 1 × PBS and 200 μL of complete culture medium.
-
200 µL of the appropriate cell suspension was added to each well to give cell numbers of 25,000, 50,000 and 1 00,000 cells/well.
-
The plates were placed directly into an incubator at 37 °C with 5 % CO2.
-
Medium changes were performed every 2 days.
3. Example Data
3.1. 12 well plate format (AVP002)
PC-3 cells were maintained on Alvetex Scaffold for up to 7 days. At specific time points, cultures were harvested and analysed by histological staining (Figure 2) and MTT viability assay (Figure 3) to monitor cell survival and proliferation within the scaffold. For histology and MTT assay protocols refer to Alvetex Protocols.
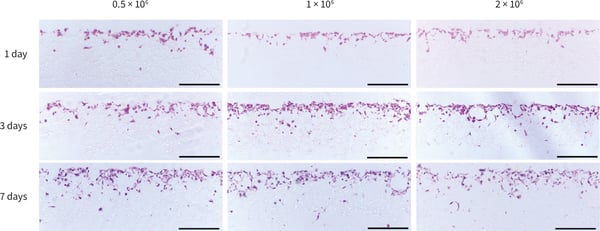
Figure 2. Histological analysis of PC-3 cells grown in Alvetex Scaffold 12-well plate format (AVP002) for up to 7 days at three different cell seeding densities. PC-3 cells grew in the top third of the scaffold with only a small number of cells migrating through the scaffold structure. Scale bars: 200 9 µm.
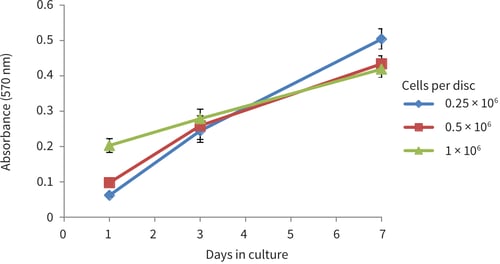
Figure 3. PC-3 growth curves obtained from MTT viability analysis for up to 7 days in Alvetex Scaffold 12-well plate format (AVP002). Cells were seeded at three different densities (0.25 × 106, 0.5 × 106 and 1 × 106 cells per disc). PC-3 cells showed good linear growth for up to 7 days. However, a drop in growth may be observed thereafter as the culture begins to approach confluency in the scaffold (n = 12, mean ± SD).
3.2. 6 well insert format (AVP004)
PC-3 cells were maintained on Alvetex Scaffold in 6 well insert format for up to 7 days. At specific time points, cultures were harvested and analysed by histological staining (Figure 4) and MTT viability assay (Figure 5) to monitor cell survival and proliferation within the scaffold. For histology and MTT assay protocols refer to Alvetex Protocols.
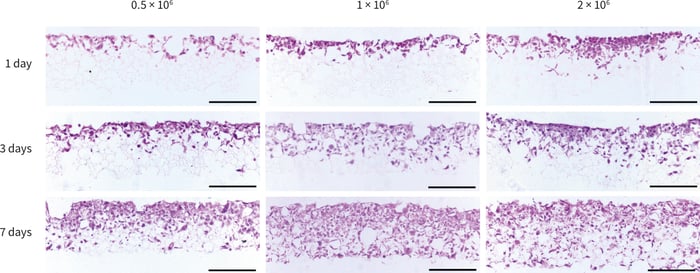
Figure 4. Histological analysis of PC-3 cells grown in Alvetex Scaffold in 6-well insert format (AVP004) for up to 7 days at three different seeding densities. Cells initially penetrated into the top third of the structure, subsequently invading downwards into the majority of the scaffold after 7 days. Scale bars 200 µm.
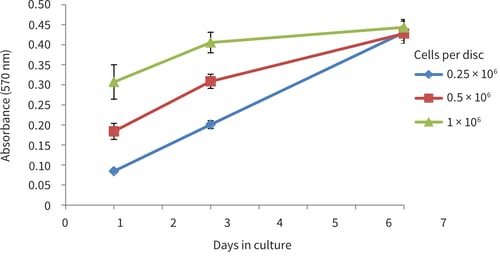
Figure 5. PC-3 growth curves obtained from MTT viability analysis for up to 7 days in Alvetex Scaffold in 6-well insert format (AVP004). Cells were seeded at three different densities (0.5 × 106, 1 × 106 and 2 × 106 cells per disc). PC-3 cells showed good linear growth for up to 7 days however a drop in growth may be observed thereafter as the culture begins to approach confluency in the scaffold (n = 9, mean ± SD).
3.3. 96 well plate format (AVP009)
PC-3 cells were maintained on Alvetex Scaffold 96 well plate format for up to 10 days. After 3 days cultures were harvested and analysed by histological staining (Figure 6) and at 1, 3, 7 and 10 days for MTT viability assay (Figure 7) to monitor cell survival and proliferation within the scaffold. For histology and MTT assay protocols refer to Alvetex Protocols.
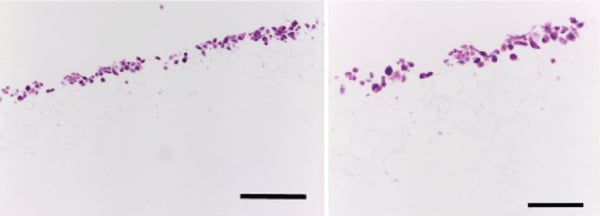
Figure 6. Histological analysis of PC-3 cells grown in Alvetex Scaffold in 96-well plate format for 3 days. Cells grew within the top third of the scaffold, and exhibited 3D cell morphology. Scale bars: Left, 200 µm; right, 100 µm.
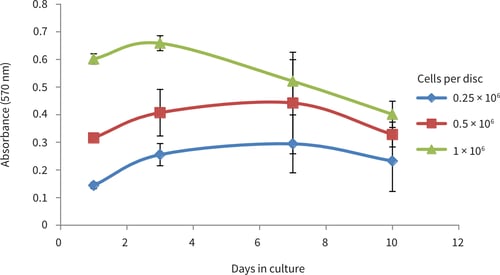
Figure 7. PC-3 prowth curves obtained from MTT viability analysis for up to 10 days in Alvetex Scaffold in 96 well plate format. PC-3 cell growth plateaued between 3 and 7 days at cell seeding densities of 25,000 and 50,000 cells/well. At a seeding density of 100,000 cells/well cell growth plateaued at 3 days and viability began to decrease thereafter (n = 6, mean ± SD).
4. References
- Kaighn ME, Naravan KS, Ohnuki Y, Lechner JF, Jones LW (1979) Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest Urol 17:16-23.
- Chlenski A, Nakashiro K, Ketels KV, Korovaitseva, GI, Oyasu R (2001) Androgen receptor expression in androgen independent prostate cancer cell lines. Prostate 47:66–75.