Pluripotent stem cells (PSC), such as embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), are a key component of the growing field of regenerative medicine. Use of culture conditions free of animal products (xeno-free) is essential for PSC projects that are destined for the clinic. NutriStem hPSC XF medium (01-0005) and iMatrix-511 (NP892-012) form an ideal system for xeno-free, feeder culture of human PSCs.
NutriStem® hPSC XF Medium
NutriStem® hPSC XF Medium is a defined, xeno-free, serum-free culture medium that is made to GMP specifications and designed to support the propagation and expansion of human ESCs and iPSCs in a feeder-free culture system. NutriStem contains low concentrations of growth factors such as FGF-basic, providing a simplified medium to maintain the cells full differentiation potential. NutriStem has been used to support a wide variety of iPS differentiation protocols, including cardiomyocytes[1], neurons[2], hepatocytes[3], and endothelial progenitor cells (EPCs)[4]. A Drug Master File for NutriStem hPSC XF medium is on file with the US FDA to support clinical applications.
 |
 |
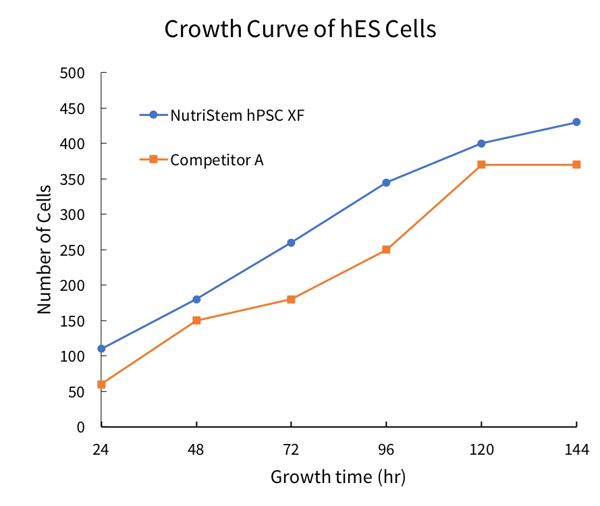
Figure 1: NutriStem® hPSC XF Medium enables excellent proliferation of undifferentiated hES and hiPS cells. (Left) Proliferation of H1 hES cells cultured in Corning Matrigel-coated 96-well plates in NutriStem XF/FF Medium and the leading competing medium for feeder-free culture. Medium was changed and proliferation was assessed every 24 hours in culture. (Right) iPSCs cultured in NutriStem hPSC XF medium
iMatrix-511
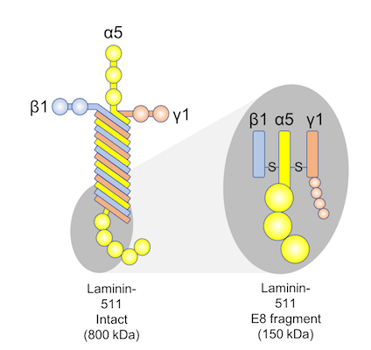
Recombinant Laminin-511 E8 Fragments is a chemically-defined, xeno-free human laminin substrate for cell growth. Laminin-511 E8 fragments have been shown to support greater adhesion of human ESCs than Corning® Matrigel®, fibronectin, or vitronectin[5], presumably through interaction with a6b1 integrin in PSCs. No karyotypic abnormalities were observed after at least 20 passages of PSCs on laminin-511 E8 fragments. iMatrix-511 supports high-efficiency reprogramming of a variety of target cells to iPSCs using REPROCELL’s Stemgent® StemRNA™ 3rd Gen Reprogramming technology (00-0076).
 |
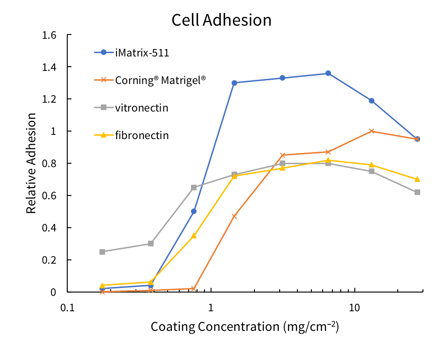 |
Figure 2: Diagram of Laminin-511 E8 fragment structure, plus graph showing adhesion of H9 ESCs to iMatrix-511 coated plates surpasses that of other popular coatings. Adhesion was measured 24 hr after plating, by washing and staining staining with crystal violet.
A powerful system for culturing human iPSCs and ESCs
Using NutriStem and iMatrix-511 together gives you a powerful system for feeder-free, xeno-free culture of human iPSCs and ESCs. Cells from skin (fibroblasts) as well blood (endothelial progenitor cells; EPCs) and urine (urine-derived epithelial cells) are readily reprogrammed using the StemRNA 3rd Gen technology (00-0076), and the iPSCs maintain pluripotency, as shown by ICC and in vitro differentiation, even after extensive passaging. This system provides a robust, xeno-free culture system for human iPSCs.
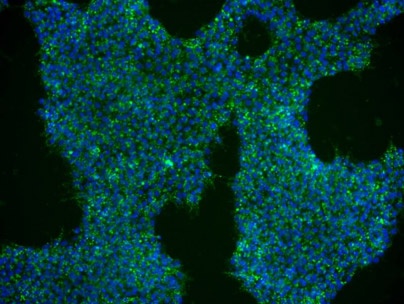 |
Figure 3: Human iPSCs derived from EPCs were cultured in NutriStem hPSC XF medium on iMatrix-511. At passage 6 the cells were stained with DAPI and anti-SSEA-4 antibody. |
 |
 |
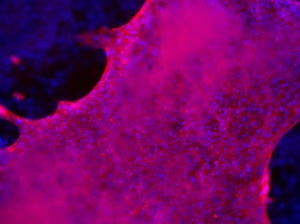 |
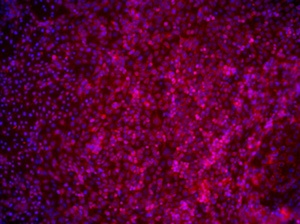
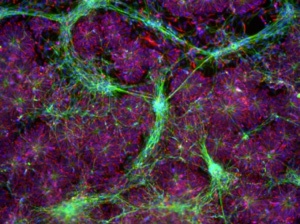
Figure 4: Human iPSCs at passage 8 in NutriStem medium on iMatrix-511 were differentiated in vitro and stained for specific markers; Left, Early endoderm: stained with DAPI (blue) and anti-AFP (red); Center, neuronal cells: stained with anti-nestin (red) and anti-β-tubulin (green); Right, Cardiomyocytes: stained with DAPI (blue) and anti-troponin T (red).
References
- Jacquet et al. Three Huntington’s disease specific mutation-carrying human embryonic stem cell lines have stable number of CAG repeats upon in vitro differentiation into cardiomyocytes PLoS ONE 10:5 (2015)
- Schwab et al. Neurite aggregation and calcium dysfunction in iPSC-derived sensory neurons with Parkinson’s disease related LRRK2 G2019S mutation Stem Cell Reports 5:6 (2015)
- Tolosa et al. Transplantation of hESC-derived hepatocytes protects mice form liver injury Stem Cell Res Therapy 6:246 (2015)
- Nguyen et al. Differentiation of human embryonic stem cells to endothelial progenitor cells on laminins in defined and xeno-free system Stem Cell Reports 7:4 (2016)
- Miyazaki et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells Nature Commun. 3:1236 (2012)
- Eminli-Meissner et al. Use of non-modified RNAs for the derivation of clinically-relevant iPS cell lines from human blood, urine and skin cells using GMP-compliant reagents (2016)



%20(1).png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs)%20(1).png)
.jpg?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).jpg)
.png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).png)

