It is well recognized that one size does not fit all when it comes to the treatment of many diseases. Getting the right drug to the right patient at the right dose has become the focus of precision medicine, which provides hope that patients may receive the most appropriate treatment sooner, improving their quality of life and reducing the support required from health care systems and wider society [1]. Health economists are recognising the potential of precision medicine and are beginning to apply the concept to their research [2].
The genomics revolution has underpinned much of this research. As the cost of gene sequencing has fallen, the ability to rapidly identify an individual’s genotype as part of routine health care has become possible. However, for precision medicines to be developed, genomics must be linked to pharmacology: relating the individuals genotype to the effectiveness, potency and tolerability of a drug. It is through pharmacogenomics that truly personalized therapies may emerge, yet the link between genomics and pharmacology may not be properly understood until expensive and risky clinical trials are conducted.
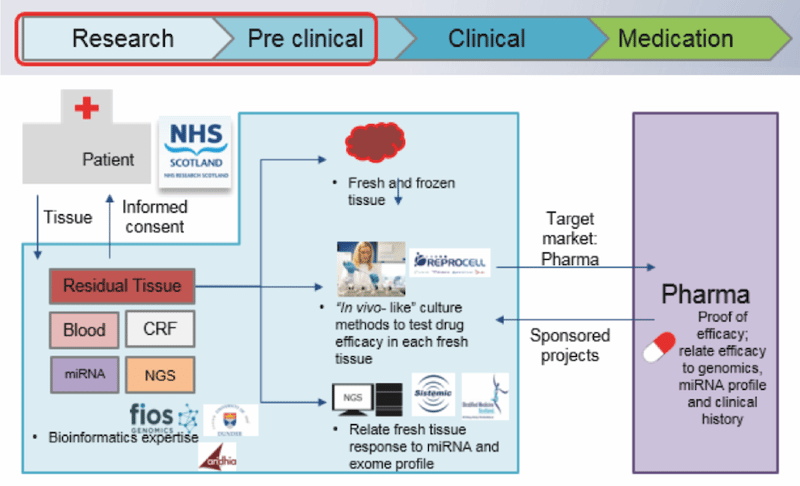
Here we describe a collaboration between industry, the National Health Service (NHS) and academia that sought to demonstrate how early understanding of both pharmacology and genomics can improve strategies for the development of precision medicines. By using the latest pharmacology techniques in human fresh tissues acquired from target patient populations, combined with genomics and clinical metadata associated with each individual, an improved understanding of the link between genetics and inter-individual drug responses emerges.
Current challenges in Drug Discovery
An early understanding of patient stratification during drug discovery is becoming increasingly important. Selection and optimization of candidate drugs for well-defined patient subsets has the potential to help in the design of more rapid, targeted clinical trials.
A key incentive to better understand pharmacogenomics during the drug discovery process is the rapid increase in drug development costs. The most recent estimates of the out-of-pocket costs (i.e. excluding capital costs) of drug development are in the region of $890m[7], with approximately 70% of the costs incurred during clinical development. The most common cause of failure is poor efficacy at phase II or III[3,4,5,6], which is in part attributed to trials of entire patient populations that include both “responders” and “non-responders”. Precision medicine can improve the prediction of clinical efficacy by selecting for clinical trials only those patient sub-populations likely to gain clear benefit; such predictions are dependent on the quality of the information used to stratify the patient sub-populations at an early stage of development. Early data on the effectiveness of drugs in different patients is essential to the development of precision medicines. Pre-clinical tests of drug effects must therefore closely reflect the patient population.
Human tissue can increase physiological relevance
The most desired traits in pre-clinical models are “physiological relevance” and the ability to translate findings to likely clinical responses[3,6,7,8], including a desire to model the likely variation in effectiveness of a new drug within the patient population. Human fresh tissues and complex 3D tissue models that reflect the biology of disease are therefore increasingly being used by Pharma to improve the prediction of efficacy in clinical trials[7,9,10]. Although the data between different patients can be variable, this is now viewed as an opportunity for an early understanding of the extent and causes of inter-patient variation in drug response.
Example of human organoculture in use
Chronic Obstructive Pulmonary Disease (COPD) is a major health problem and is an example of a complex condition, with many clinical phenotypes. Many patients receive minimal clinical benefit from common medications, most likely due to the combination of variations in disease subtype and genotype. The clinical variation in drug response is apparent in ex vivo pharmacology experiments using fresh lung tissues [11].
REPROCELL (Biopta) used diseased tissue ethically acquired from patients suffering from COPD to investigate inter-patient variability in drug efficacy using ex vivo organocultures of fresh lung tissue as the test system. In order to assess patient variation and responsiveness to both ‘standard of care’ and potential novel therapies, the reduction in inflammatory cytokines in the presence of various test drugs was used as the measure of drug efficacy. The individual patient responses were then matched against genotype and microRNA profiles in an attempt to identify unique predictors of drug responsiveness and demonstrate the combined power of pharmacology and genomics during pre-clinical development.
A full description of the results has now been published in PLOS One.
References
- Schork. Time for one-person trials. Nature Comment 520: 609-611 (2015)
- Biankin et al. Therapeutic development in precision oncology. Nature 15:361-70 (2015)
- Cook et al. Lessons learned from the fate of AstraZeneca’s drug pipeline: a five-dimensional framework. Nature Review Drug Discovery 13:419-31 (2014)
- LaVallee. Found in Translation. European Pharmaceutical Review 6:43-45 (2012)
- Ashgar et al. Engineering cancer microenvironments for in vitro 3-D tumor models. Materials Today. 18:10 (2015)
- Zanoni et al. 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Scientific Reports 11:19103 (2016)
- Kauffman et al. Microarray data quality control improves the detection of differentially expressed genes. Genomics 95: 138-42 (2010)
- Zheng. Statistical Prediction of HLA Alleles and Related Analysis in Genome-Wide Association Studies. Biostatistics PhD Thesis, University of Washington (2013)
- Purcell et al. PLINK: a toolset for whole-genome association and population-based linkage analysis. American Journal of Human Genetics; 81 (2007).
- Johnson et al. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 8:118-127 (2007)
- Brandsma et al. Differential effects of fluticasone on extracellular matrix production by airway and parenchymal fibroblasts in severe COPD. American Journal of Physiology-Lung Cellular and Molecular Physiology 305:8 (2013)


.jpg?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).jpg)
.png?width=1000&height=562&name=New%20Approach%20Methodologies%20(NAMs).png)