Poly-D or L-lysine Coating of Alvetex® Scaffold
● Download this protocol as a PDF (1.7 KB)
1. Introduction
The following protocol outlines how to coat Alvetex Scaffold membranes with poly-D or Llysine in order to facilitate cell attachment and migration within the scaffold.
Poly-L-lysine is a synthetic amino acid that has been widely used as a coating agent in tissue culture to enhance cell attachment to untreated plastic and glass. When Poly-L-lysine is applied, it renders the material positively charged and increases the electrostatic attraction between the surface and the negatively charged cells thus improving adhesion. The use of Poly-D-lysine is favoured with cells displaying high proteolytic activity as it is less prone to breakdown than the naturally-occurring L-enantiomer. Otherwise, Poly-D-Lysine and Poly-L-Lysine are equivalent for these purposes. Refer to manufacturer’s instructions when choosing poly-lysine formats and dilutions required for the desired cell type.
Note also that the available molecular weight (MW) ranges of poly-lysine vary. The lower the MW, the less viscous the solution, while the higher the MW, the more binding sites per molecule. In this protocol 3T3 fibroblasts were grown on poly-L-lysine (Sigma-Aldrich, P4707) coated Alvetex Scaffold in 12-well plate (AVP002) format for 7 days. The MW of poly-L-lysine ranged from 70-150 kDa, which remained easy to handle while providing sufficient binding sites for cell attachment.
2. Method
- Prepare Alvetex Scaffold for coating by first treating with 70 % ethanol followed by two PBS washes as described in the relevant product information leaflet. Leave Alvetex Scaffold in the second PBS wash until ready to apply the poly-L-lysine solution.
- Aspirate the second PBS wash and add 500 μl of poly-D or L-lysine per well. Replace plate lids and leave to stand for 1 hour at room temperature.
- Tilt the 12-well plate and gently aspirate any excess fluid from the edge of the wells. If using well insert Alvetex Scaffold formats, remove excess fluid from Alvetex Scaffold by gently tapping the plate or Petri dish on the worktop. Check that no residual fluid is hanging from the base of the well inserts. Aspirate to remove any residual coating agent from the bottom of the wells.
- Prepare cells for seeding in the appropriate culture media and seed directly on the wet poly- L-lysine coated Alvetex Scaffold membrane in the volumes relevant for the Alvetex Scaffold product format. Allow the cells to settle for 30-90 minutes in an incubator (5 % CO2, 37 °C) before flooding with media.
3. Example: Growth of 3T3 Cells in poly-L-lysine Coated Alvetex Scaffold
3.1. Cell Culture details
3T3 cells were routinely maintained in T-75 flasks. Complete media consisted of: DMEM media supplemented with 10 % v/v FBS, 2 mM L-glutamine and 100 U/ml Penicillin/Streptomycin. Alvetex Scaffold 12-well plates (AVP002) were coated in poly-L-lysine (Sigma-Aldrich, P4707, MW: 70-150 kDa, 0.01 % solution) as described above.
Cells were seeded at a density of 0.5 × 106 cells in 125 μL media suspension per disc and were left to settle for 60 minutes in an incubator (5 % CO2, 37 °C). Media was carefully added to each sample (9 mL per well). Cultures were maintained for 7 days, with media changes every second day.
3.2. Results
Pre-coating of Alvetex Scaffold discs with poly-L-lysine resulted in enhanced infiltration of 3T3 cells into the scaffold compared with control cultures in uncoated Alvetex Scaffold. Cells were seen to reside deep within the scaffold and in greater abundance after 7 days of growth in treated discs. These findings indicate that pre-treatment of Alvetex Scaffold with well established tissue culture coating agents is able to enhance the attachment and growth of appropriate cell types into the 3D structure.
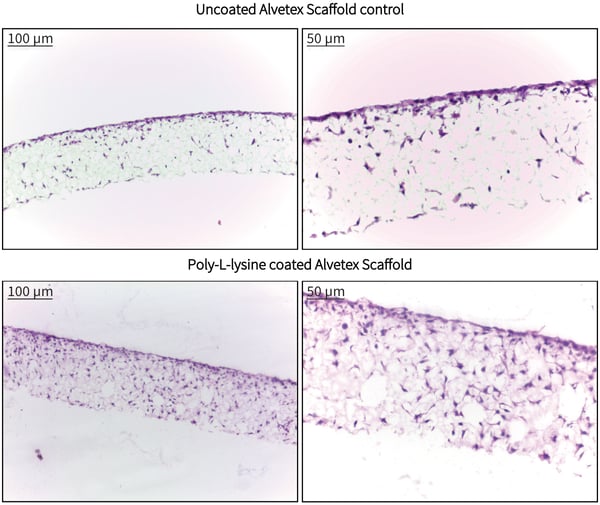
Figure 1. Brightfield micrographs showing the structure of 3T3 cells cultured on non-coated and poly-L-lysine coated Alvetex Scaffold discs in 12 well plate format (AVP002) after 7 days culture.