Stemolecule™ Doxycycline hyclate
04-0016
Brand: Stemolecule™
Doxycycline hyclate (dox) is a broad spectrum antibiotic derivative of tetracycline and an inhibitor of matrix metalloproteinases.
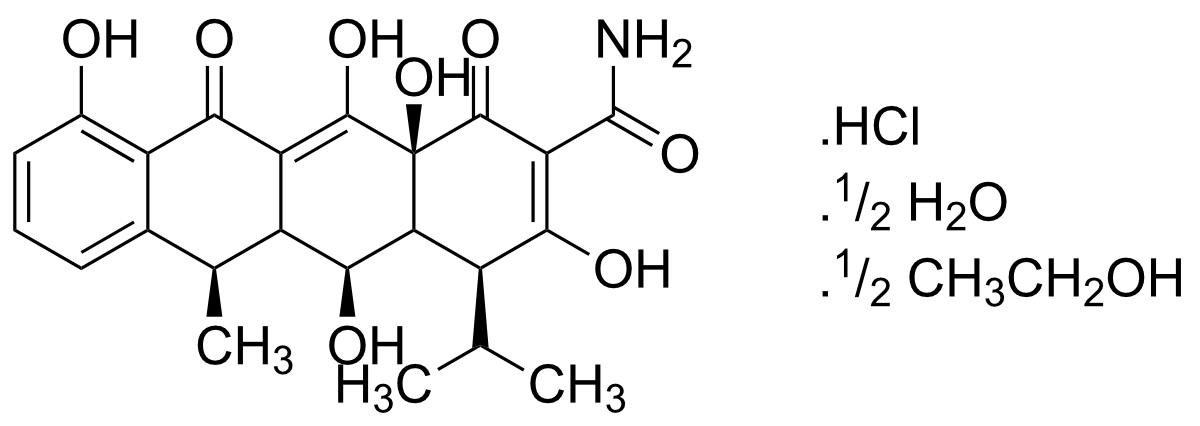
- (2Z,4S,5S,6R,12aS)-2-[amino(hydroxy)methylidene]-4-(dimethylamino)-5,10,11,12a-tetrahydroxy-6-methyl-4a,5,5a,6-tetrahydro-4H-tetracene-1,3,12-trione; ethanol; hydrochloride
Currency:
| Product name | Catalog number | Pack size | Price | Price (USD) | Price (GBP) | Price (EUR) |
|---|---|---|---|---|---|---|
| Stemolecule™ Doxycycline hyclate | 04-0016 | 10 mg | (select above) | $ 70.00 | £ 58.00 | € 68.00 |
Note: prices shown do not include shipping and handling charges. To get a quote, make an inquiry.
Product Information
Stemolecule Doxycycline hyclate (dox) is a broad spectrum antibiotic derivative of tetracycline and an inhibitor of matrix metalloproteinases1. Tetracycline-controlled transcriptional activation is a method of inducible expression whereby transcription is reversibly turned on or off in the presence of tetracycline or one of its derivatives such as dox2. For this type of reprogramming method, dox-inducible lentiviral reagents are used to induce the expression of virally transduced genes and generate induced pluripotent stem (iPS) cells from somatic cells3-7.
This product is intended for Research Use Only.

Stemgent and the Stemolecule brand name are trademarks of REPROCELL Inc., Japan.
Product Name: Stemolecule Doxycycline Hyclate
Catalog Number: 04-0016
Size: 10 mg
Alternate Name(s): (2Z,4S,5S,6R,12aS)-2-[amino(hydroxy)methylidene]-4-(dimethylamino)-5,10,11,12a-tetrahydroxy-6-methyl-4a,5,5a,6-tetrahydro-4H-tetracene-1,3,12-trione; ethanol; hydrochloride
Chemical Formula: C22H24N2O8· HCl · ½(H2O) · ½(C2H6O)
Molecular Weight: 512.94
CAS Number: 24390-14-5
Purity: Greater than 98% by TLC analysis
Formulation: Yellow to yellow with a green cast powder
Solubility: For a 10 mM stock solution of doxycycline hyclate, reconstitute the compound by adding 1.95 mL of water to the entire contents of the vial.
Storage and Stability: Store powder at 4 °C protected from light. Information about the stability of Stemolecules in solution is largely not available. As a general guideline, we recommend that stock solution be freshly made and stored in aliquots at −20 °C, protected from light. The effect of storage of stock solutions should be verified for each application.
Quality Control: The purity of doxycycline hyclate was determined by TLC analysis. The accurate mass was determined by mass spectrometry. Cellular toxicity of doxycycline hyclate was tested on mouse embryonic stem cells.
Specification Sheets:
Safety Data Sheets:
- Burgraff, D., Trinkl, A., Dichgans, M., and Hamann, G.F. Doxycycline inhibits MMPs via modulation of plasminogen activators in focal cerebral ischemia. Neurobiol Dis 25: 506-513 (2007).
- Bujard, H., and Gossen, M. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci 89: 5547-5551 (1992).
- Brambrink, T., Foreman, R., Welstead, G.G., Lengner, C.J., Wernig, M., Suh, H., and Jaenisch, R. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell 2: 151-159(2008).
- Wernig, M., Lengner, C.J., Hanna, J., Lodato, M.A., Steine, E., Foreman, R., Staerk, J., Markoulaki, S., and Jaenisch, R. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nat Biotechnol 26: 916-924 (2008).
- Hockemeyer, D., Soldner, F., Cook, E.G., Gao, Q., Mitalipova, M., and Jaenisch R. A drug-inducible system for direct reprogramming of human somatic cells to pluripotency. Cell Stem Cell 3: 346-353 (2008).
- Welstead, G.G., Brambrink, T., and Jaenisch, R. Generating iPS cells from MEFS through forced expression of Sox-2, Oct-4, c-Myc, and Klf4. J Vis Exp 7: 734 (2008).
- Markoulaki, S., Hanna, J., Beard, C., Carey, B.W., Cheng, A.W., Lengner, C.J., Dausman, J.A., Fu, D., Gao, Q., Wu, S., Cassady, J.P., and Jaenisch, R. Transgenic mice with defined combinations of drug-inducible reprogramming factors. Nat Biotechnol 27: 169-171 (2009).